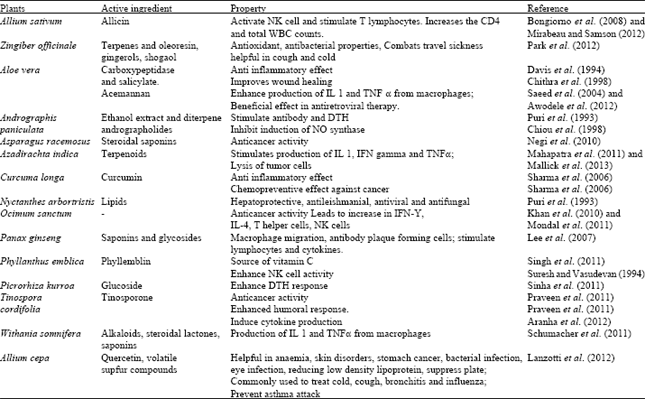
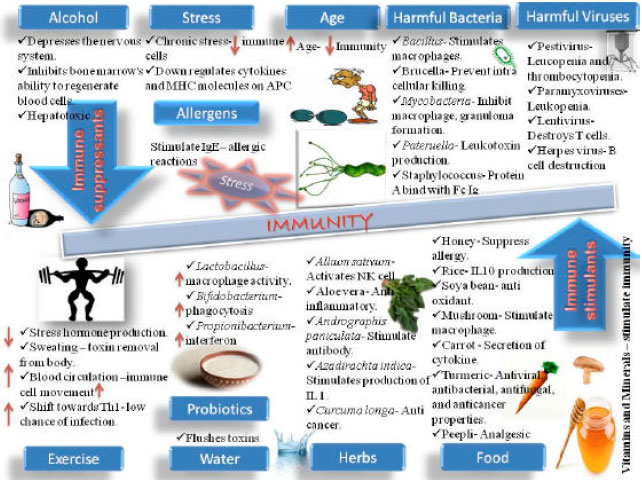
Review Article
Immunomodulators in Day to Day Life: A Review
Department of Animal Nutrition, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Viswavidyalaya Evum Go-Anusandhan Sansthan, Mathura-281001, India
Abhijeet M. Ingle
Division of Bacteriology and Mycology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India
Amit Kumar Verma
Department of Veterinary Epidemiology and Preventive Medicine, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Viswavidyalaya Evum Go-Anusandhan Sansthan, Mathura-281001, India
Ruchi Tiwari
Department of Veterinary Microbiology and Immunology, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Viswavidyalaya Evum Go-Anusandhan Sansthan, Mathura-281001, India
K. Karthik
Division of Bacteriology and Mycology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India
Sandip Chakraborty
Deaprtment of Animal Resource Development, Pt. Nehru Complex, Agartala, Tripura-799001, India
Rajib Deb
Animal Genetics and Breeding, Project Directorate on Cattle, Indian Council of Agricultural Research, Grass Farm Road, Meerut, (UP)-250001, India
S. Rajagunalan
Division of Veterinary Public Health, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India
Rajesh Rathore
Division of Bacteriology and Mycology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India
Kuldeep Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP)-243122, India