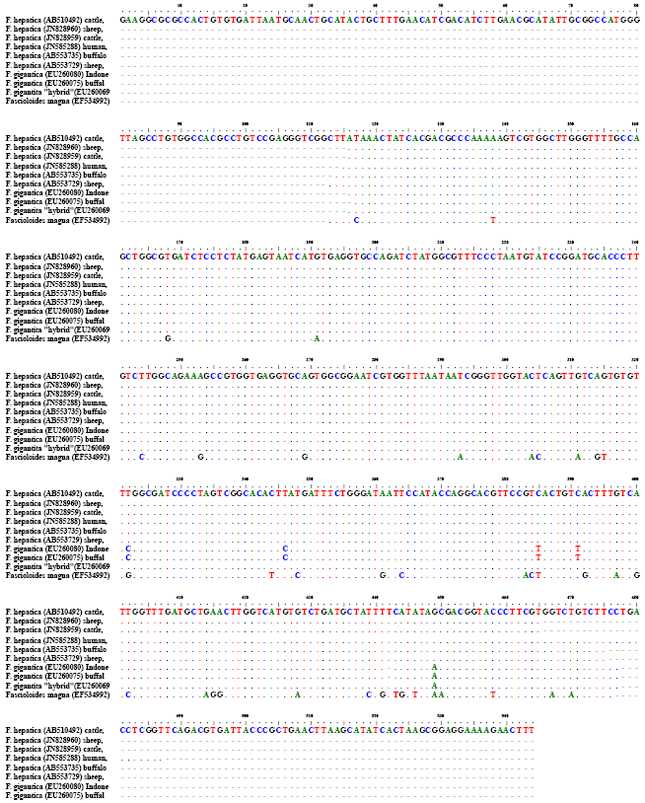
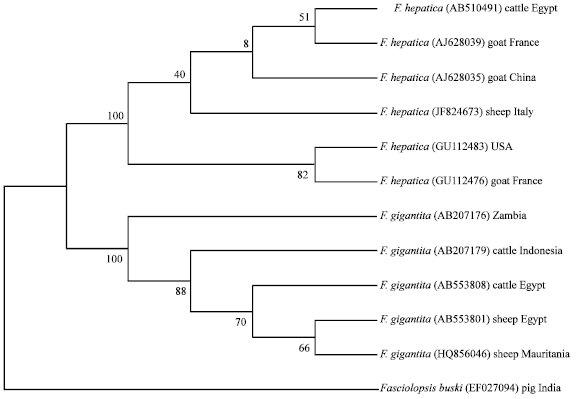
Research Article
Molecular and Phylogenetic Status of Fasciola sp., of Cattle in Qena, Upper Egypt
Department of Veterinary Parasitology, Faculty of Veterinary Medicine, South-Valley University, 83523 Qena, Egypt
Asmaa M. Metwally
Department of Veterinary Parasitology, Faculty of Veterinary Medicine, South-Valley University, 83523 Qena, Egypt
Khaled Sultan
Department of Parasitology, Faculty of Veterinary Medicine, Kafrelsheikh University, 33516, Kafr El-Sheikh, Egypt