Research Article
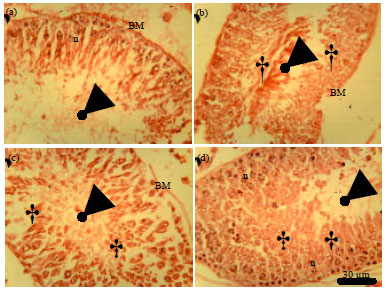
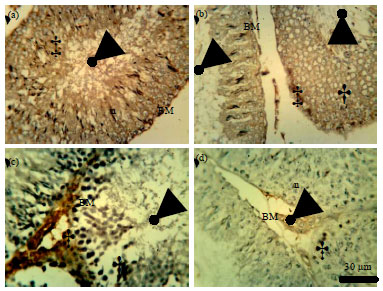
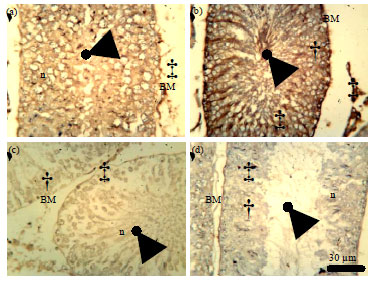
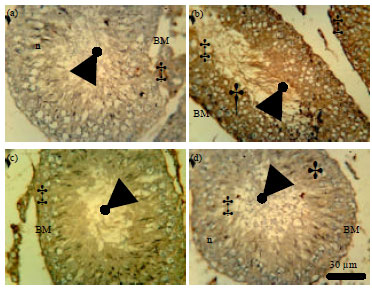
Role of Se+Zn in Regeneration (Ki-67) Following Pb Toxicity (p53andcad) in the Germinal Epithelium of Adult Wistar Rats
Department of Anatomy, College of Medicine and Health Sciences, Osun State University, Oshogbo, Nigeria
O.M. Ogundele
Department of Anatomy, College of Medicine and Health Sciences, Afe Babalola University, Ado-Ekiti, Nigeria
F.I. Duru
Department of Anatomy, College of Medicine and Health Sciences, University of Lagos, Lagos Sate, Nigeria
A.A. Oshinubi
Department of Anatomy, College of Medicine and Health Sciences, University of Lagos, Lagos Sate, Nigeria
D.T. Falode
Department of Nursing, College of Medicine and Health Sciences, Afe Babalola University, Ado-Ekiti, Nigeria