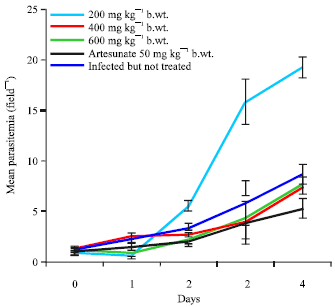
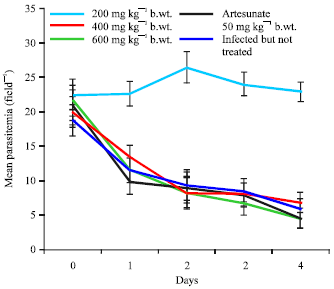
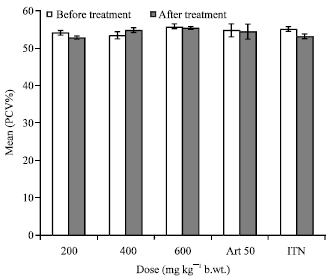
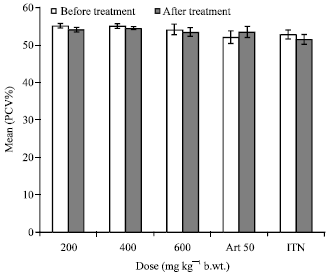
Research Article
Evaluation of Haematological Changes in Plasmodium-berghei-infected Mice Administered with Aqueous Extract of Phyllantus amarus
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
Abdulkadir
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
A. Timothy A. Gbodi
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
U. M. Bello
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
Hussaini Anthony Makun
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
D. J. Amah
Trypanosomiasis and Malaria Research Unit, Department of Biochemistry, Federal University of Technology, Minna, Nigeria
E. O. Ogbadoyi
Global Institute for Bio-Exploration (GIBEX), Federal University of Technology, Minna, Nigeria