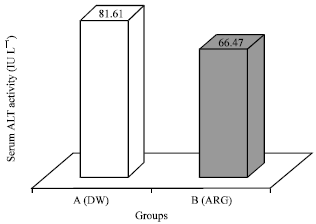
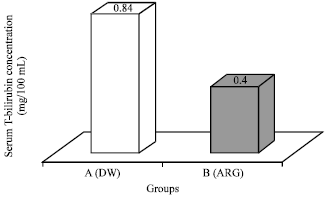
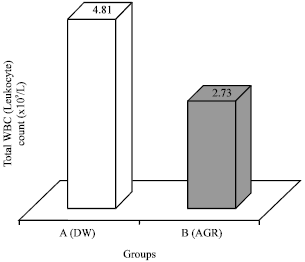
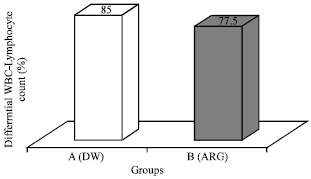
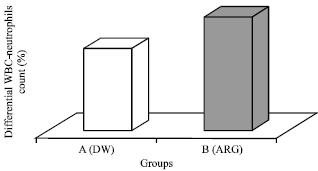
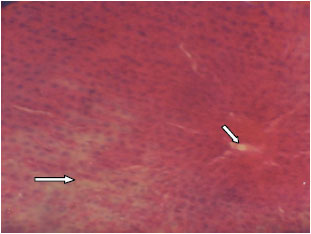
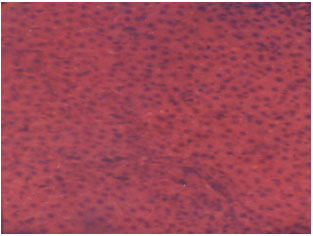
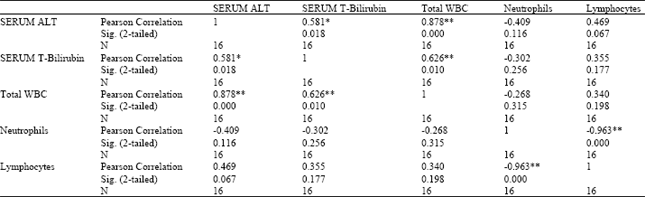
Research Article
Alterations in the Liver Histology and Markers of Metabolic Syndrome Associated with Inflammation and Liver Damage in L-arginine Exposed Female Wistar Albino Rats
Department of Biochemistry, University of Nigeria Nsukka, Enugu State, Nigeria
L.U.S. Ezeanyika
Department of Biochemistry, University of Nigeria Nsukka, Enugu State, Nigeria
I.I. Ijeh
Department of Biochemistry, Michael Okpara University of Agriculture Umudike, Abia State, Nigeria