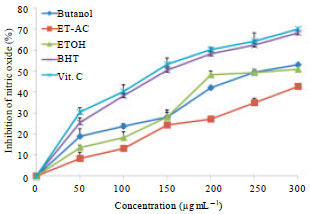
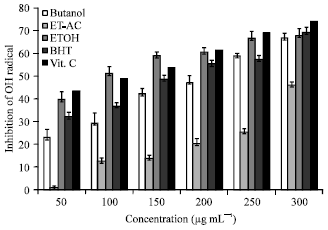
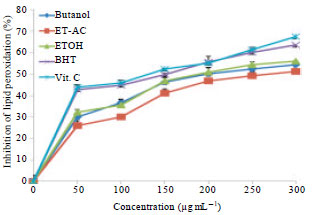
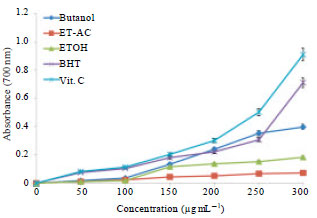
Research Article
Polyphenolic Contents and Free Radical Scavenging Potential of Extracts from Leaves of Ocimum americanum L.
Department of Biochemistry, University of Ado Ekiti, Nigeria
O.I. Oloyede
Department of Biochemistry, University of Ado Ekiti, Nigeria
A.J. Afolayan
Department of Botany, University of Fort Hare, Alice, South Africa