Research Article
Evaluation of the Effect of Processing Methods on the Nutritional and Anti-nutritional Compositions of Two Under-utilized Nigerian Grain Legumes
Department of Food Science and Engineering, Ladoke Akintola University Ogbomoso, Nigeria
S.S. Sobowale
Department of Food Technology, Moshood Abiola Polytechnic, Abeokuta, Nigeria
G.O. Ogunlakin
Department of Food Science and Engineering, Ladoke Akintola University Ogbomoso, Nigeria









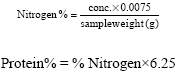
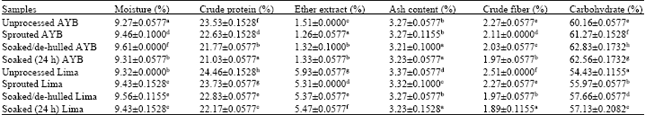
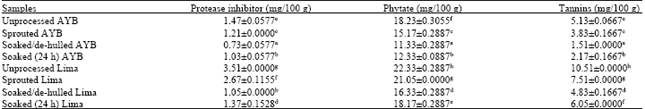
AMAH Gogonte Hezekiah Reply
The source cited for condensed tannin determination (Burns, 1971), referenced as Burns, R.E., 1971. Method for estimation of tannin in grain sorghum. Agron. J., 63: 511-512, should be cross checked for year of publication. I think the work of Burns on method for condensed tannin determination in sorghum was first published in 1902. Please, the purpose for this comment was to guide against transmitting the error and nothing else