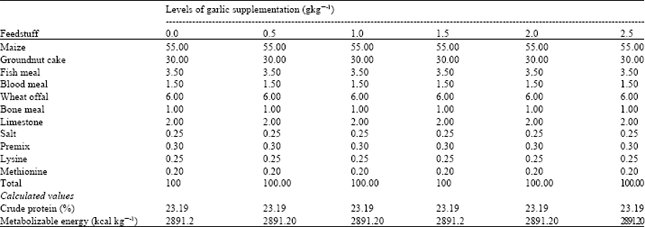
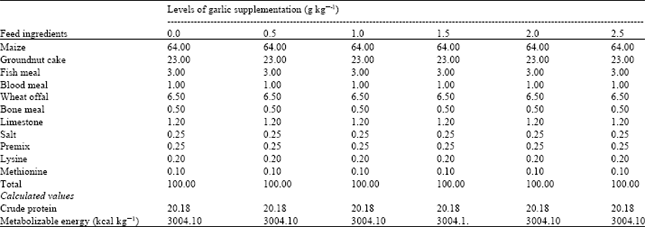
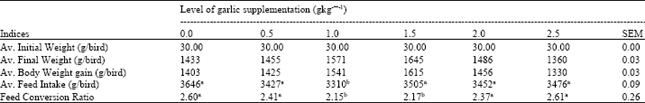
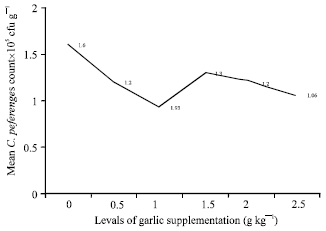
Research Article
In vivo Antimicrobial Potentials of Garlic against Clostridium perfringens and Its Promotant Effects on Performance of Broiler Chickens
Department of Theriogenology and Animal Production, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria
E.B. Ibitoye
Department of Theriogenology and Animal Production, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria
Y.U. Dabai
Department of Veterinary Microbiology, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria
S. Garba
Veteinary Teaching Hospital, Faculty of Veterinary Medicine, Usmanu Danfodiyo University Sokoto, Nigeria