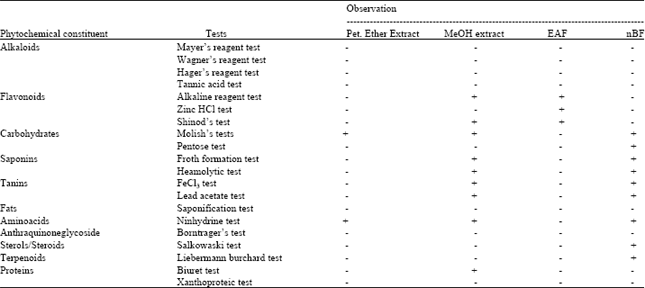
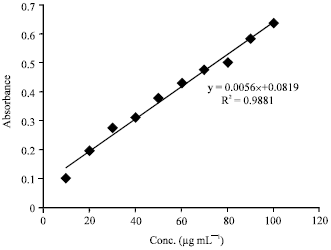
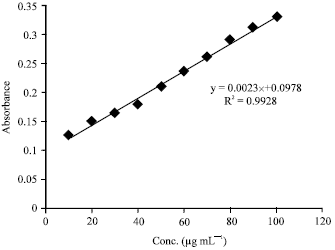
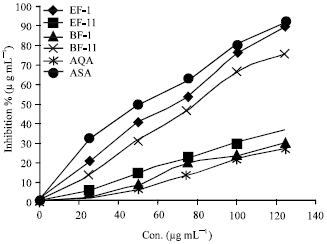
Research Article
Phytochemical Screening and In-vitro Antioxidant Activity Isolated Bioactive Compounds from Tridax procumbens Linn
P.G. Department of Botany, Govt. Maharaja Autonomous P.G. College, Chhatarpur-471405, M.P, India
Abrar Hussain Mir
Pest Control and Ayurvedic Drug Research Laboratory, S.S.L. Jain P.G. College, Vidisha-464001, M.P. India
Manik Sharma
P.G. Department of Zoology, Bhoj Mahavidhyalya Bhopal M.P, India
Mohd Yousuf Malla
P.G. Department of Zoology, Bhoj Mahavidhyalya Bhopal M.P, India
Sumeerah Qureshi
Pest Control and Ayurvedic Drug Research Laboratory, S.S.L. Jain P.G. College, Vidisha-464001, M.P. India
Mohd Iqbal Mir
P.G. Department of Zoology, Bhoj Mahavidhyalya Bhopal M.P, India
Yogesh Chaturvedi
P.G. Department of Botany, Govt. Maharaja Autonomous P.G. College, Chhatarpur-471405, M.P, India