Research Article
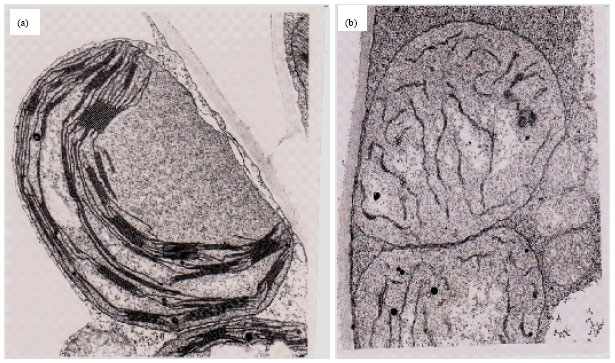
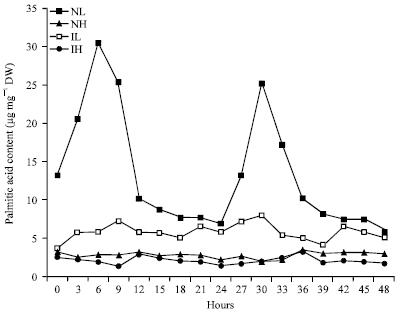
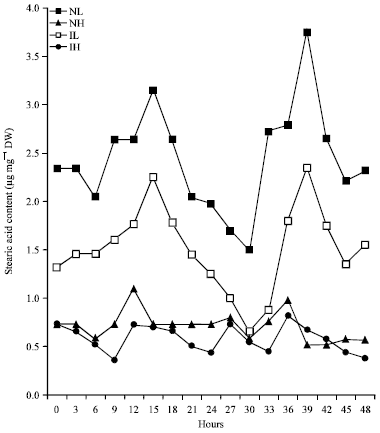
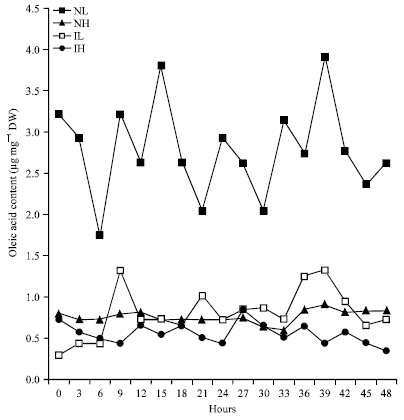
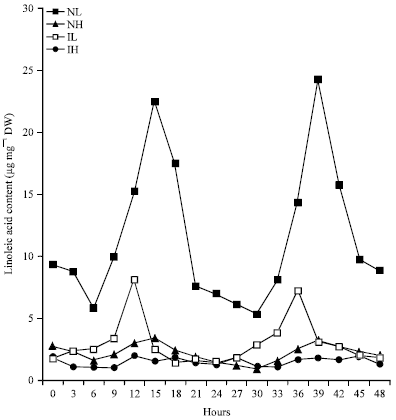
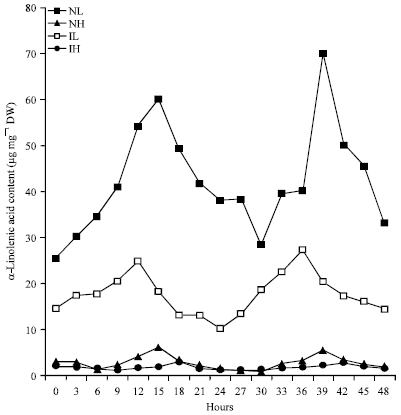
Rhythmic Changes in the Levels of Fatty Acids in Leaves of Phaseolus aureus Seedlings did not Tightly Depend upon High/Low Temperatures Cycles and Alterations in Chloroplast Ultrastructure
Department of Plant Biology, Faculty of Sciences, University of Dschang, P.O.Box 67, Dschang, Cameroon