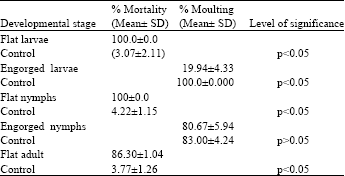
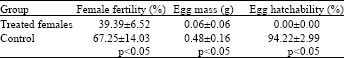
Research Article
Metarhizium anisopliae as a Biological Control Agent Against Hyalomma anatolicum (Acari: Ixodidae)
Veterinary Research Institute, P.O. Box 8067, Alamarat, Khartoum, Sudan
M. T. Shigidi
Faculty of Veterinary Medicine, University of Khartoum. P.O. Box 32, Khartoum North, Sudan
S. M. Hassan
Faculty of Veterinary Medicine, University of Khartoum. P.O. Box 32, Khartoum North, Sudan