Research Article
Immunomodulatory Activities of Yoyo Bitters: Recommended Dose Precipitated Inflammatory Responses in Male Wistar Rats
Department of Biochemistry, Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology, P.M.B. 4000, Ogbomoso, Oyo State, Nigeria
A. Adetutu
Department of Biochemistry, Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology, P.M.B. 4000, Ogbomoso, Oyo State, Nigeria
J. A. Adebisi
Department of Biochemistry, Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology, P.M.B. 4000, Ogbomoso, Oyo State, Nigeria












Taiwo Mary Morenike Reply
Please, what are the side effects of yoyo bitters?
Dr. Oyewo E. Bukoye
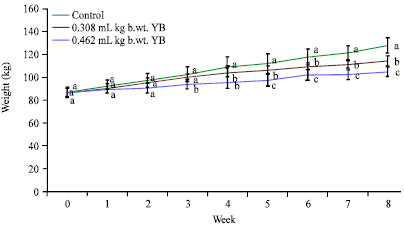
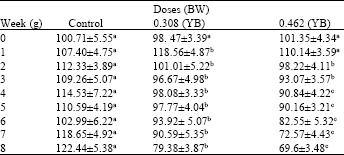
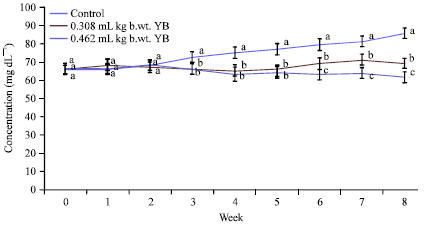
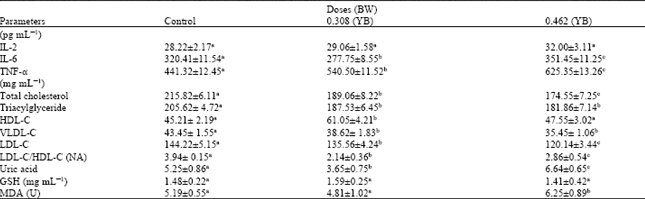
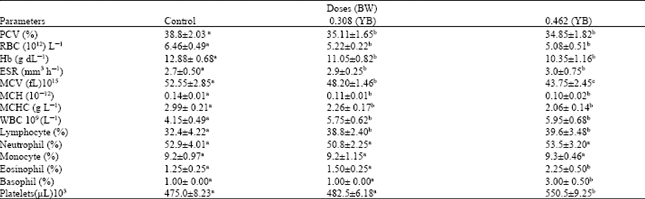
Yoyo bitters on long time usage at the recommended indicated the induction of oxidative stress and inflammatory responses, which are the key causes of many degenerative diseases. The daily consumption of this bitters is discouraged.