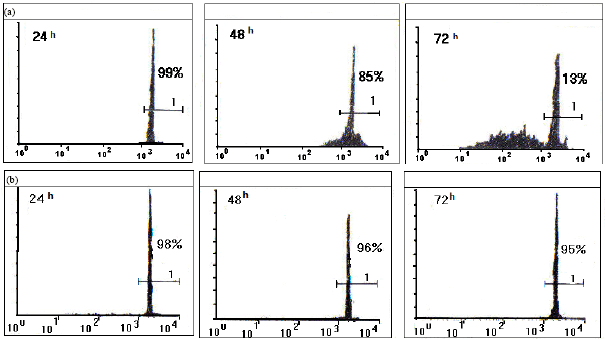
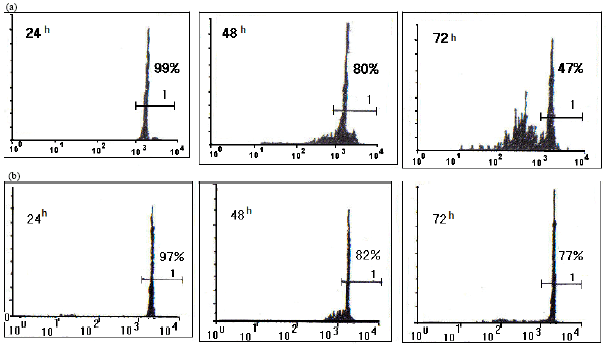
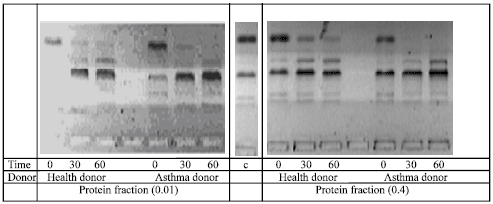
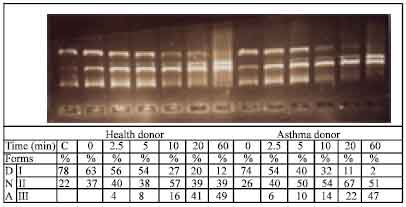
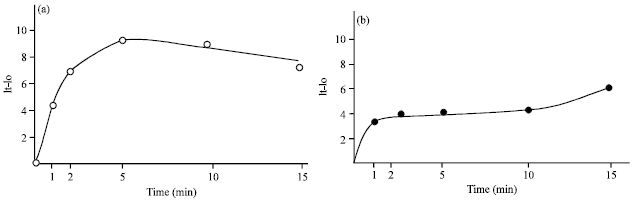
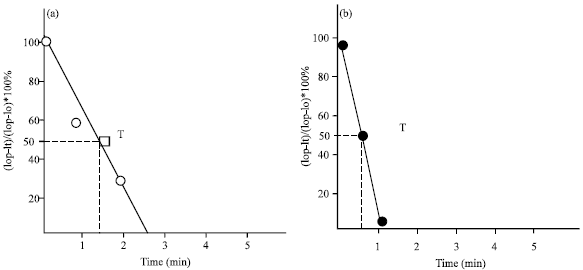
Research Article
The Particularities of Protein fraction in the Apoptosis of Lymphocytes of Patients with Asthma
Laboratory Acid Nucleic, Department of Biochemistry, Faculty of Biology, Kazan Federal University, Kremlyovskaya str, 18 Kazan 480008, Republic of Tatarstan, Russian Federation
Z. I. Abramova
Laboratory Acid Nucleic, Department of Biochemistry, Faculty of Biology, Kazan Federal University, Kremlyovskaya str, 18 Kazan 480008, Republic of Tatarstan, Russian Federation
N. Aikou
Laboratoire de biologie et de typage moleculaire en microbiologie, Departement de biochimie et biologie cellulaire, Faculte des Sciences et Techniques (FAST), Universite d`Abomey-Calavi (UAC-Benin), 05PB1604 Cotonou, Benin
A. Sezan
Laboratoire de biologie et de typage moleculaire en microbiologie, Departement de biochimie et biologie cellulaire, Faculte des Sciences et Techniques (FAST), Universite d`Abomey-Calavi (UAC-Benin), 05PB1604 Cotonou, Benin
S. O. Kotchoni
Department of Biology and Center for Computational and Integrative Biology, Rutgers University, Camden, NJ 08102, USA
S. A. Akpona
Laboratoire de Biochimie et Biologie Cellulaire, Faculte de Medecine, Universite de Parakou, BP:123, Parakou, Benin
Lamine Baba-Moussa
Laboratoire de biologie et de typage moleculaire en microbiologie, Departement de biochimie et biologie cellulaire, Faculte des Sciences et Techniques (FAST), Universite d`Abomey-Calavi (UAC-Benin), 05PB1604 Cotonou, Benin