Review Article
In vitro Propagation of Ginger (Zingiber officinale Rosc.) through Direct Organogenesis: A Review
Department of Crop Science, Faculty of Agriculture, Eastern University, Chenkalady, Sri Lanka
INTRODUCTION
Ginger (Zingiber officinale Rosc.) of the family Zingiberaceae is an important herbaceous plant primarily used as a spice and in medicine at the global level. Underground rhizomes are utilized in herbal medicine, food and beverage and form an important raw material in pharmaceutical industries (Bhagyalakshmi and Singh, 1988). Fresh and dried rhizomes are dealt in the world as well as it becomes an important as a value-added product. China and India are the two major ginger growing countries (Ravindran and Babu, 2005) and Sri Lanka produces relatively small quantity of ginger than these two countries in 2010 (FAOSTAT, 2013). It is cultivated in different ecological regions of Sri Lanka it is mainly grown in wet and intermediate zones. Sri Lanka produced 10,780 metric tons of ginger from the total harvested extent of 1,890 ha in 2009 (Central Bank, 2012). In Sri Lanka, different varieties of ginger are cultivated as a monocrop in home garden and as an intercrop (Abeysekera et al., 2005). Even though ginger is grown throughout the country, the required amount of ginger is not supplied to meet the local demand. Hence, a major portion of ginger is being importing from other countries. Such a situation is enforcing to increase the production of ginger in Sri Lanka. As conventional propagation has a low rate of multiplication, in vitro culture technology is an alternative method for in vitro multiplication of ginger for commercial cultivation. This review aims to emphasize the clonal propagation of ginger through organogenesis under in vitro conditions for the rapid production of healthy plantlets.
Ginger is highly valuable monocotyledonous plant having underground rhizome for its aromatic and medicinal properties. It contains high quantitative secondary metabolite, Oleoresin (Sakamura et al., 1986). Ginger is a native to tropical South East Asia and later introduced to other tropical regions of the world (Pieris, 1982). It is commercially cultivated as an annual and it is being used as a spice and in medicine in Asia (Pandey et al., 1997). Ginger grows well on loam soil incorporated with humus but it does not stand under water logging conditions (Purseglove, 1976). Under favourable conditions, the ginger plants continuously survive through rhizomes and the cultivated ginger is not known to set seeds (Ravindran et al., 2005). The axilliary and terminal buds grow up as leafy stem which is known as an aerial stem. Leaves are sheathing and arranged alternatively. The structure of rhizome consists of two different zones which are separated by intermediate layers and a cambium-like layer presented is a significant trait in ginger (Ravindran et al., 1998).
Ginger is commercially obtainable in various forms and the processed products are in powder, oil and oleoresin forms (Kizhakkayil and Sasikumar, 2009). The rhizome is valued for its flavour and contains the essential oils and oleoresins (Balladin et al., 1998). The rhizomes are internationally commercialized for use in food and pharmaceutical industries. The more volatile oleoresin is accountable for the pungent flavour which is a source of antioxidants (Balachandran et al., 2006). Ginger is also known as a remedy for disorders of the gastrointestinal tract and recommended for use in cardiopathy and high blood pressure in South Asia by the conventional healers (Ghayur et al., 2005). Further, ginger rhizome taken with salt before meals can purify the tongue and it is supposed to enhance the appetite and makes an acceptable sensation (Nadkarni, 1998). Shukla and Singh (2007) claimed that ginger rhizome shows cancer preventive property.
Generally, ginger is vegetatively propagated through mature rhizome segments having three to four buds. It has the habit of rare flowering and non-viable seed production (Ikeda and Tanabe, 1989; Malamung et al., 1991). The rhizome produces an average of 12 lateral buds per season (Bhagyalakshmi and Singh, 1988) and it is a slow process. Seed rhizome sprouts during the favourable climatic conditions. In addition, ginger cultivation is threatened by systemic diseases that are spread through infected rhizomes when used as planting materials. Mostly the pre-emergence rot and yellow diseases are transmitted from infected rhizomes (Dohroo, 1989) and 50-70% cost of production goes to planting materials (Villamor, 2010). Ginger rhizomes are used for human consumption and also for other commercial purposes but a greater portion of total yield are needed as planting unit in traditional cultivation of ginger. The increase of rhizome production is practicable using efficient control of diseases (Hosoki and Sagawa, 1977; Balachandran et al., 1990). Poor seed setting is a major obstacle in crop improvement of ginger. It is therefore necessary to find the alternative source of disease free planting materials to establish large scale production.
In vitro culture technology is a significant role in rapid propagation of healthy planting materials. This technology may be appropriate to multiply ginger rhizomes for production due to the following reasons: (1) To obtain disease free planting materials, (2) To overcome dormancy problem in seed rhizomes and (3) To obtain planting materials to cultivate under favourable conditions (Hiremath, 2006). High rate of in vitro plant regeneration from cultured tissues is a pre-requisite for successful application of tissue culture technique for crop improvement (Akter, 2001). Ginger is a monocotyledonous crop and required number of disease free plantlets can be acquired from a single in vitro cultured explant within a year. Hence, in vitro multiplication is essential for improvement of ginger (Kambaska and Santilata, 2009). This technique has been used as a tool for commercial propagation of many plant species successfully (George, 2003).
IN VITRO STUDIES IN GINGER
In vitro culture technique is an efficient method for rapid propagation of important plant species and their germplasm conservation. Each plant cell has the capacity to progress into a complete plant in plant tissue culture (Haberlandt, 1902) and it is achieved with the discovery of auxins and cytokinins (Hiremath, 2006). In vitro plant regeneration of axilliary and adventitious shoots from shoot tips has been attempted by callus culture (Hosoki and Sagawa, 1977). However, organogenesis and somatic embryogenesis through callus culture are not recommended for clonal propagation. Genetic uniformity is sustained to a large degree in shoot tips and axillary bud cultures (Evans et al., 1981). Clonal propagation methods through meristem tip culture have been reported (Pillai and Kumar, 1982; Smith and Hamill, 1996; Rout et al., 2001) to obtain pathogen free plant materials for commercial utilization and also removal of nematodes in ginger by in vitro culture technique has been stated (De Lange et al., 1987).
With regard to in vitro culture of ginger, there are reports on micropropagation from shoot tips and rhizome buds (Smith and Hamill, 1996; Khatun et al., 2003; Kambaska and Santilata, 2009; Hamirah et al., 2007; Sathyagowri and Seran, 2011a), adventitious shoot organogenesis from shoot tips (Inden et al.,1988; Pandey et al., 1997), somatic embryogenesis (Babu et al., 1992; Kackar et al., 1993), in vitro germplasm conservation (Dekkers et al., 1991), encapsulation of disease-free buds (Sharma et al., 1994) and the production of microrhizomes (Sharma and Singh, 1995). Thus, application of tissue culture technique overcomes the problems associated with propagation and production of high yielding cultivars of ginger. The major role of in vitro culture is to conserve the genetic variation and plant populations of ecologically and commercially viable genotypes in order to prevent their possible destruction (Kambaska and Santilata, 2009).
IN VITRO MICROPROPAGATION
Micropropagation of ginger using rhizome bud explants has been reported by several researchers (Hosoki and Sagawa, 1977; Bhagyalakshmi and Singh, 1988; Balachandran et al., 1990; Dogra et al., 1994; Sharma and Singh, 1997; Rout et al., 2001; Kavyashree, 2009; Sathyagowri and Seran, 2011a). In addition to rhizome buds, aerial stem explants have also been used for multiple shoot initiation (Lincy et al., 2004) as well as in vitro plant regeneration (Ikeda and Tanabe, 1989; Lincy and Sasikumar, 2010). Vegetative buds are commonly used as the explants however rhizome explants often transmit soil-borne diseases and they are not available during the crop season (Lincy and Sasikumar, 2010). In conventional vegetative propagation, multiplication rate is slow and it limits the availability of good quality superior planting materials for ginger cultivation. As a result in vitro micropropagation technique is the best method to supply disease free plant propagules continuously for commercial use.
Careful selection of variables including the source, type, developmental stage and size of explants, sterilization of explants, growth regulator and medium, culture conditions etc. is an important for successful application of in vitro technology. The optimization of these factors guides to successful tissue cultures in many plant species (Xiao et al., 1997). All the tissues or organs are not equally competent to show morphogenesis (Hartamann et al., 1997). In micropropagation technique, aim of culture establishment is to place explants into aseptic culture under in vitro culture environments for constant shoot production and critical phases are choice of plant materials, sterilization of explants and nutrient medium (Hartamann et al., 1997). The above aspects should be considered for the initial culture establishment and fast clonal propagation of ginger under aseptic conditions. The key industrial goal of in vitro culture is to produce more number of plants in months (Haque et al., 2009).
Explant source: The physiological state of a plant varies naturally according to the developmental stages of plants and changes of environmental conditions. Active shoot tips and axilliary buds are mostly used on micropropagation of plant species. If rhizome explants of ginger are less active or dormant it may require different levels of hormones during initial culture establishment to induce sprouting of buds for shoot formation. Generally, contamination of field grown rhizome is the persistent problems under in vitro conditions since rhizome contact with the soil therefore it should be disinfected carefully. To reduce the level of contamination, rhizome explants could be obtained from ginger plants grown under glasshouse conditions. Explants from healthy, vigorous plants are more suitable to produce successful in vitro cultures.
Explant type: The different types of explants used in micropropagation of ginger and other related species are meristem, axilliary buds, shoot tips and aerial pseudostems. However, rhizome buds and shoot tips are commonly used as explants and they are the responsive explants for micropropagation of pathogen free propagules on a large scale (Kambaska and Santilata, 2009; Lincy and Sasikumar, 2010). This is with the findings of Olivier (1996) that axillary buds are the most appropriate explants for the effective clonal propagation of ginger. Further, sprouting buds of turmeric rhizomes are suitable materials for vegetative propagation under in vitro conditions (Kuruvinshetti and Iyer, 1981; Shetty et al., 1982) as compared with dormant buds which have to force for shoot initiation. Several scientists (Balachandran et al., 1990; Smith and Hamill, 1996; Pandey et al., 1997; Kambaska and Santilata, 2009; Naz et al., 2009; Behera et al., 2010) have been able to produce plantlets easily using sprouting bud explants of ginger and turmeric. Leafy aerial pseudostems and crown segments of ginger have been also successfully cultured under aseptic conditions (Ikeda and Tanabe, 1989) and meristem tip culture is very useful for the elimination of viruses from infected plant materials.
Explant size: An optimum size of explants requires initiating tissue cultures. Very small explants are less transmitting endogenous infestation. On the other hand the smaller explants are more likely to be damaged during handling and do not last well in initial culture but big explants may be difficult to purify effectively (George et al., 1996). Several workers have been used different sized explants of ginger as shown in Table 1. The meristems with the apical dome or with leaf primordia (0.1 to 0.5 mm) are used as explants (Rout et al., 2001). Small shoot tips exhibits slow initial growth because of low nutrients in explants but less risk of contamination on micropropagation of ginger (Razdan, 1993; Dey and Harborne, 1997; Sathyagowri and Seran, 2011a). The size of rhizome explants has an influence on shoot bud initiation and morphogenic response under in vitro conditions. Shoot tips of 0.2-0.9 mm long is optimum for in vitro propagation (Huang, 1995). This is in agreement with the findings of Sathyagowri and Seran (2011a) that the best size of the rhizome buds is 0.5 cm long for in vitro culture initiation and shoot multiplication of ginger among the different sizes (0.5, 1.0 and 2 cm long) of explants tested.
Explant sterilization: As rhizome explants are underground part of ginger plant the in-vitro establishment of clean culture is not easy due to the presence of pathogens in these living materials. Several researchers have been reported on the surface sterilization of rhizome ginger.
| Table 1: | Studies on in vitro propagation of ginger directly from rhizome explants |
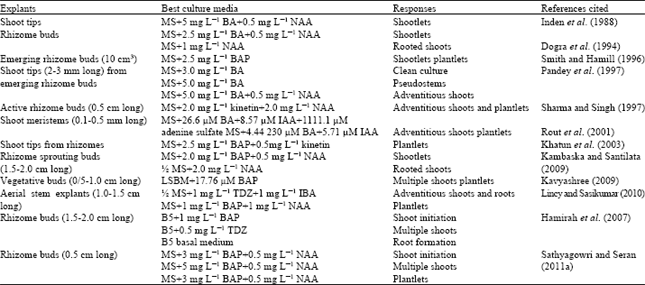 | |
| B5: Gamborg et al. (1968) basal medium, LSMB: Linsmaier and Skoog’ basal medium, MS: Murashige and Skoog (1962) basal medium | |
Sterilization requires the use of chemicals that are toxic to microorganisms but non-toxic to plant materials. The type, concentration and exposure time of disinfectants depend on the nature of explants and plant species (Razdan, 1993). Explants are disinfected with a sterilant solution of appropriate concentration for a precise time. The commonly used disinfectants are ethanol, sodium hypochlorite and mercuric chloride. Sterilization of rhizome buds have been done using 0.1% mercuric chloride (HgCl2) solution for 10-20 min to establish aseptic cultures in ginger (Sharma and Singh, 1997; Khatun et al., 2003; Kambaska and Santilata, 2009), turmeric (Rahman et al., 2004; Bharalee et al., 2005) and black thorn (Rahman et al., 2005). This is strengthened by Pillai and Kumar (1982) who states that the shoot tips of ginger could be effectively disinfected with 0.1% HgCl2 solution and 90% ethanol. Rout et al. (2001) developed a disinfection protocol for sprouting buds of ginger with 2% (v/v) Teepol for 15 min and then with 0.2% (w/v) aqueous solution of HgCl2. In some instance, underground rhizome buds are sterilized using both suitable detergents and sterilizing agent because of high degree of bacterial and fungal contamination.
In general, commercial bleach (CloroxTM) is often used to disinfectant for surface sterilization of plant materials and also it is not hazardous. The disinfection method for emerging buds of ginger is soaked in 0.2% Tetracycline and 0.2% Metalaxyl for one and half hours before being treated with 10% Clorox for 15 min followed by 5% Clorox for 10 min (Pandey et al., 1997). The effectiveness of Clorox is related to exposure time and the rate of contamination commonly decreases with an increase in both soaking time and concentration to certain limit (Seran et al., 2007). The optimum regime for decontamination of ginger is sterilizing rhizome buds using 20% Clorox for 20 min (Hamirah et al., 2007; Sathyagowri and Seran, 2011a). However, bacterial contamination is a common problem in ginger but it can be controlled by incorporation of antibiotics to initial culture medium. In general, lack of browning and absence of contamination on in vitro explants are as the criteria for survival of explants for subsequent shoot multiplication.
Culture medium: MS (Murashige and Skoog, 1962) basal medium is most frequently used in micropropagation of herbaceous plant species. B5 (Gamborg et al., 1968) and other media are also being used by many researchers. The basic nutrient medium without growth regulators but with sucrose is generally used for initial culture of explants. Plant growth regulators mainly cytokinins and auxins are very important on micropropagation technology. Cytokinins induce to form the lateral buds from apical dominance (Wickson and Thimmann, 1958). The cell division and differentiation are influenced by auxins and cytokinins (Skoog and Miller, 1957). The culture establishment and proliferation of shoot tips and rhizome explants and their subsequent plantlet production are mostly obtained on MS basal medium containing BAP alone or in combination with NAA in ginger as given in Table 1. The effect of BAP on organogenesis and somatic embryogenesis of ginger has been reported by several researchers.
Sucrose (3%) is commonly used as a carbon source for in vitro plant propagation of ginger (Pandey et al., 1997; Smith and Hamill, 1996; Khatun et al., 2003; Hamirah et al., 2007; Sathyagowri and Seran, 2011a). This is supported by Rout et al. (2001) who states that sucrose assists to form rhizomes than other carbohydrate tested.
| Table 2: | Studies on in vitro micropropagation of turmeric (Curcuma sp.) plants belonging to the family of Zingiberaceae |
 | |
| MS: Murashige and Skoog (1962) basal medium, WPM: Woody plant basal medium | |
In culture medium, agar is used as solidifying agent ranging from 0.5, 0.8 and 0.5% by Khatun et al. (2003); 0.62% by Pandey et al. (1997); 0.8% by Sharma and Singh (1997); Smith and Hamill (1996); Sathyagowri and Seran (2011a) and the pH of medium is adjusted to 5.8 before autoclaving using 0.1 M NaOH or 0.1 M HCl.
Culture establishment: Culture medium without growth regulators fails to stimulate the shoot bud initiation in the explants after four weeks of culture but with growth regulators, bud initiation has been observed within about 7 days of culture (Kambaska and Santilata, 2009). MS media containing 0-5 mg L-1 cytokinin (BAP or kinetin) alone or with auxin (NAA or IAA or IBA) are commonly used to produce multiple shoots and their subsequent plantlet formation in ginger (Table 1) and turmeric (Table 2). BA alone or in combination with NAA is more effective for shoot and root differentiation and widely used on micropropagation (Pandey et al., 1997). This is strengthened by Sathyagowri and Seran (2011b) states that BAP (1-2 mg L-1) with 0.1 mg L-1 auxins (NAA or IAA) directs to initiate shoot buds and roots for organogenesis but BAP alone shows the potential for somatic embryogenesis when the aerial stem explants are cultured on MS medium plus BAP only or with NAA or IAA. BAP alone exhibits comparatively lower response of explants in the culture medium than BAP with NAA (Kambaska and Santilata, 2009). As culture medium is significant aspect on micropropagation of plant species, the culture environments such as light and temperature are also important factors. The cultures are generally incubated under photoperiod regime of 16 h light and 8 h dark with cool, white fluorescent light, 60-70% relative humidity and temperature of 22±2°C (Lincy and Sasikumar, 2010) or 25±2°C in culture room (Rout et al., 2001; Kambaska and Santilata, 2009; Hamirah et al., 2007).
Shoot multiplication: Cytokinin induces cell division and bud proliferation resulting in shoot formation (Lydiane and Kleyn, 1996). BAP at highest dosage is more stimulatory to shoot growth than the lowest dosage in the culture medium (Zuraida et al., 2011). In contrary, Kambaska and Santilata (2009) indicates that BAP (2.0 mg L-1)+NAA (0.5 mg L-1) gives best response for shoot multiplication (7.5 shoots per rhizome bud) but higher concentration of BAP (2.5-3.0 mg L-1) with NAA (0.5 mg L-1) exhibits callus formation with fewer number of shoots. In vitro response of rhizome buds depends on cultivars, manipulation of growth regulators, culture conditions etc. Small quantity of cytokinin may be produced by the apical rhizome buds grown under in vitro conditions (Ammirato, 1986).
The shoot proliferation mainly depends on kind and concentration of cytokinins used in culture medium. The literature survey (Table 1) reveals the optimal concentration of growth regulars for shoot multiplication of ginger. The best response (7.0-7.5 shoots per explants) of ginger for shoot multiplication is obtained on MS medium containing Kinetin or BAP with NAA (Sharma and Singh, 1997; Kambaska and Santilata, 2009). The highest number (5.33) of shoots produces on MS with 5 mg L-1 BA and 0.5 mg L-1 NAA after 5 weeks of culture (Pandey et al., 1997) and this medium gives good response for shoot proliferation (Inden et al., 1988; Choi and Kim, 1991; Sathyagowri and Seran, 2011a). However, shoot multiplication decreases with increasing dosage of BA from 6 to 8 mg L-1 in ginger (Rout et al., 1997). This finding is in conformity with Naz et al. (2009) who reports that the higher concentration of BAP decreases the shoot multiplication rate in turmeric among BAP (1-6 mg L-1) tested.
Though, BAP and kinetin are commonly used cytokinins on micropropagation TDZ has also been tested for shoot proliferation and shoot elongation. TDZ at 0.5 mg L-1 induces higher number (8) of shoots among the cytokinins even at lower concentration (Hamirah et al., 2007). However, TDZ inhibits shoot proliferation especially in solid medium (Huetteman and Preece, 1993; Amutha et al., 2006). The plantlets in the solid medium supplemented with TDZ exhibits stunted appearance but this adverse effect on shoot proliferation can be changed by using liquid medium (Lincy and Sasikumar, 2010). This is in confirmation with Mohamed et al. (2006) who states that the physiological nature of explants is altered by liquid culture.
Further, vertical sectioning the bud explant into two haves may be able to double the number of explants than using the whole bud to increase the production (Hamirah et al., 2007; Sathyagowri and Seran, 2011b). It has been reported on micropropagation of pineapple in which the shoot multiplication rate improves remarkably (De Almeida et al., 2002). Normally, culture medium for initial establishment, shoot multiplication and plantlet formation is same but the concentrations of cytokinins may vary. Cytokinin proportion is commonly increased in medium for shoot multiplication to produce microshoots. This stage is repeated until a required number of shoots is produced. Long-term in vitro culture of ginger shoots upto 1 year did not affect the shoot multiplication rate (Inden et al., 1988).
Shoot elongation and rooting: Literature survey reveals that in vitro plantlets having normal shoots and roots have been mostly obtained on MS medium supplemented with BAP (1-5 mg L-1) alone or in combination with low concentration (0.5 mg L-1) of NAA (Table 1). Addition of BAP (2.0 mg L-1) with NAA 0.5 mg L-1 exhibits to better response of ginger over BAP alone (Hashim et al., 1988; Noguchi and Yamakawa, 1998). In some cases, after shoot multiplication, shootlets are placed to second medium for shoot elongation (Sathyagowri and Seran, 2011a) or for rooting of microshoots (Dogra et al., 1994; Kambaska and Santilata, 2009; Hamirah et al., 2007). These results are in accordance with those of Inden et al. (1988) who reports that high concentrations of growth regulators reduces shoot elongation and rooting on micropropagation.
Rooting of microrhizome is generally spontaneous in ginger however it is induced on culture medium without or with auxins either NAA or IBA or IAA. Bhagyalakshmi and Singh (1988) states that IBA is more effective when compared to NAA for root formation in meristem culture. In contrary, NAA has more effective than IBA in induction of rooting (Kambaska and Santilata, 2009). In turmeric, the maximum rooting to multiple shoots is noted on half strength of MS medium with 0.5 mg L-1 NAA (Dipti et al., 2005). Rooting is best on 0.5 mg L-1 IBA but rooting of microrhizomes does not take place in the absence of IBA (Sit and Tiwari, 1998). Plantlet production with profuse rooting in vitro is an important for successful establishment of in vitro plantlets in soil (Sharma and Singh, 1995).
Acclimatization: The greenhouse and field conditions are characterized by very high light intensity and low humidity but environments in culture containers are low light intensity and very high humidity (Desjardins et al., 1987). Hence, in vitro plantlets should be carefully transferred to ex vitro conditions and field due to the changes in environmental conditions and also morphology of plantlets. Several workers reported that in vitro plantlets of ginger are effectively acclimatized and survived under field conditions (Rout et al., 2001; Khatun et al., 2003). Samsudeen et al. (2004) states that more than 80% success is achieved when plants transplanted in a potting mixture of soil, sand and vermiculite (1:1:1) and maintained in a humid chamber. in vitro plantlets survive more under ex vitro conditions when they are acclimatized under high RH and CO2 enrichment conditions (Cha-um et al., 2005).
The survival of in vitro regenerated plants to ex vitro conditions depends on the conditions during transfer of in vitro plantlets and soil substrates. During acclimatization, the survival rate of in vitro tea plants in soil only is more than in the potting mixture of sand:soil (1:1) and plant survival may be increased by the addition of moisture absorbing material to soil (Seran et al., 2005). Coir dust absorbs and holds soil water for a longer time and gives a good physical support for the in vitro plantlets (Gangopadhaya et al., 2002). It is locally available material and it can be incorporated into a potting mixture to enhance moisture content of soil mixture for better plant growth (Seran et al., 2011).
PLANT PRODUCTION
Rooting in-vitro is not preferred due to labour-intensive and costly (Hazarika, 2003) and this procedure is costing about 35 to 75% of micropropagation (Debergh and Maene, 1981). However, labour costs can be reduced considerably if rooting occurs ex vitro (Hazarika, 2003). Studies revealed that about 0.5 million clones could be produced in half a year through subculture and also rhizomes from the micropropagated clones is not rot during storage (Sharma and Singh, 1997).
Ginger (Zingiber officinale Rosc.) is an important aromatic and medicinal plant. It is traditionally propagated through rhizome segments. As a result of low multiplication rate in conventional propagation, micropropagation technology is the alternative method to supply healthy plant propagules continuously for commercial use. With regard to tissue culture of ginger, there are reports on successful micropropagation from rhizome buds. An optimum size of sterilized explants and suitable culture medium mainly require initiating in vitro cultures. MS basal media supplemented with 0-5 mg L-1 cytokinin (BAP or kinetin) alone or with auxin (NAA or IAA or IBA) are commonly used to produce in vitro plantlets in ginger. The shoot multiplication mainly depends on type and strength of cytokinins incorporated in nutrient medium. Rooting of microrhizome is generally spontaneous in ginger but auxins either NAA or IBA or IAA may be used for root formation. Several workers reveal that in vitro plantlets of ginger are effectively acclimatized and survived under field conditions. Perhaps, this technique may be useful for rapid clonal propagation of healthy ginger plants within a short period for commercial production.
ACKNOWLEDGMENT
The author is grateful to Miss S. Sathyagowri, Department of Crop Science, Faculty of Agriculture, Eastern University of Sri Lanka for her support given to collect the relevant materials for the preparation of this study.