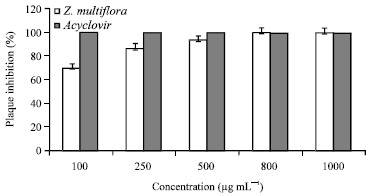
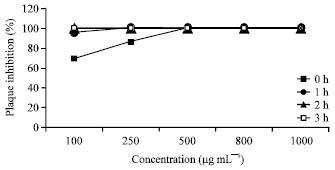
Research Article
Anti Herpes simplex-1 Activity of A Standard Extract of Zataria multiflora Boiss
Department of Microbiology, Faculty of Medicine, Kerman University of Medical Sciences, Kerman, Iran
Mehdi Ansari-Dogaheh
Pharmaceutics Research Center, Faculty of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
Fariba Sharififar
Herbal and Traditional Medicines Research Center, Kerman University of Medical Sciences, Kerman, Iran
Mojtaba Shakibaie
Herbal and Traditional Medicines Research Center, Kerman University of Medical Sciences, Kerman, Iran
Mahnaz Heidarbeigi
Herbal and Traditional Medicines Research Center, Kerman University of Medical Sciences, Kerman, Iran