Research Article
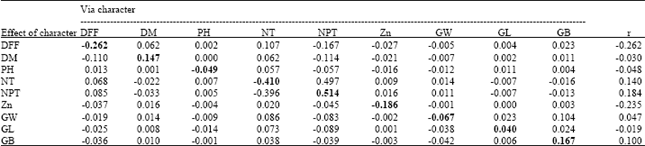
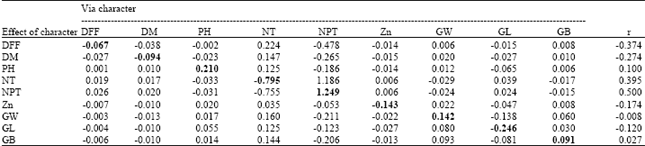
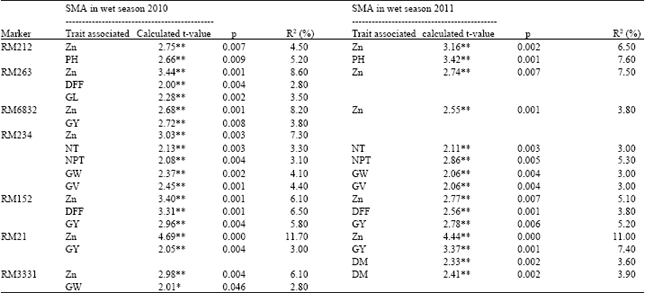
Genetic Evaluation of Recombinant Inbred Lines of Rice (Oryza sativa L.) for Grain Zinc Concentrations, Yield Related Traits and Identification of Associated SSR Markers
Department of Plant Biotechnology, University of Agricultural Sciences, GKVK, Bangalore-560065, India
G.K. Naveen
Department of Plant Biotechnology, University of Agricultural Sciences, GKVK, Bangalore-560065, India
S. Rakhi
Department of Plant Biotechnology, University of Agricultural Sciences, GKVK, Bangalore-560065, India
H.E. Shashidhar
Department of Plant Biotechnology, University of Agricultural Sciences, GKVK, Bangalore-560065, India