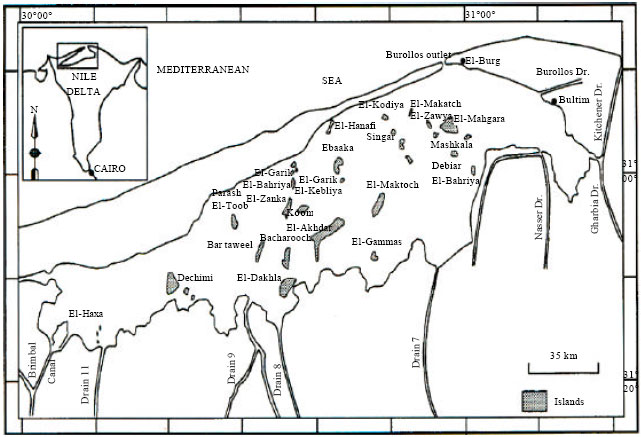
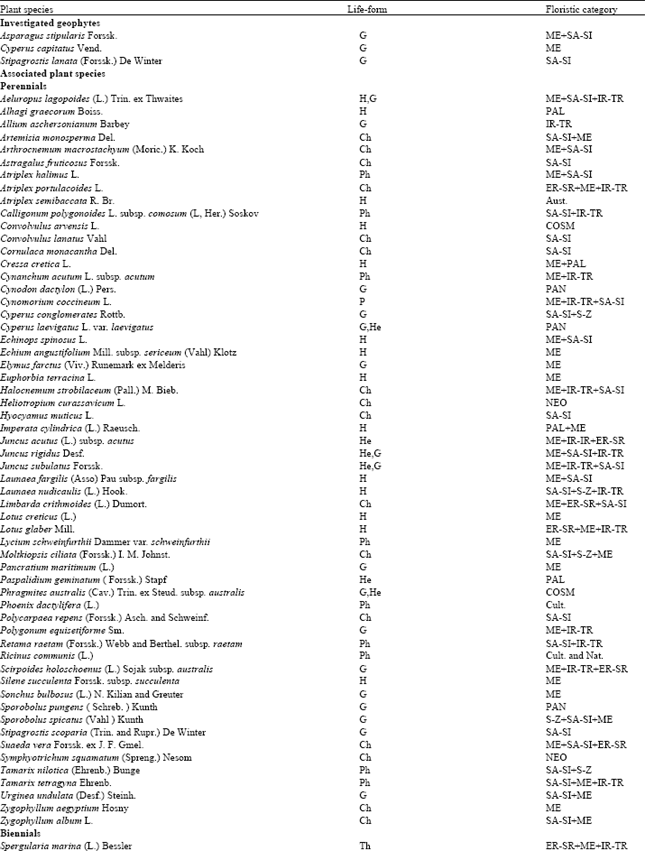
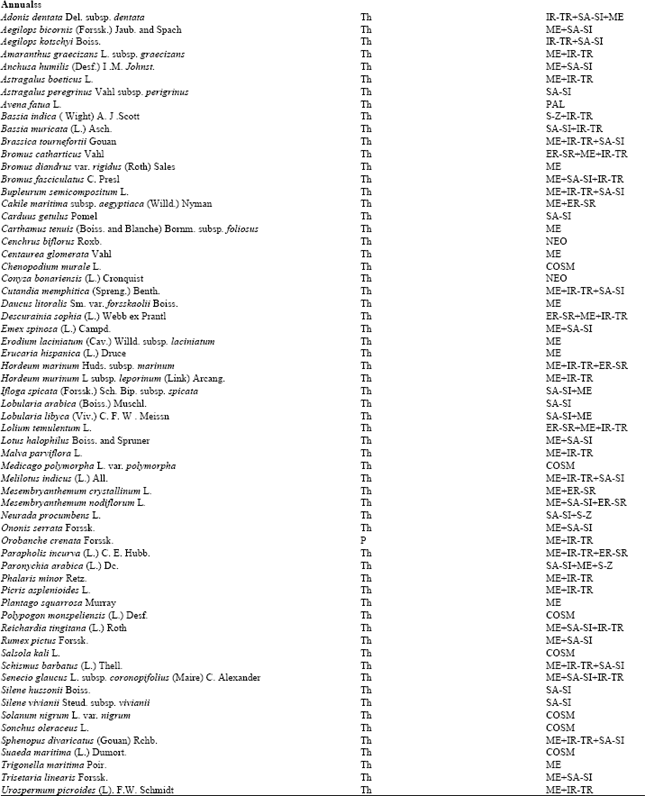
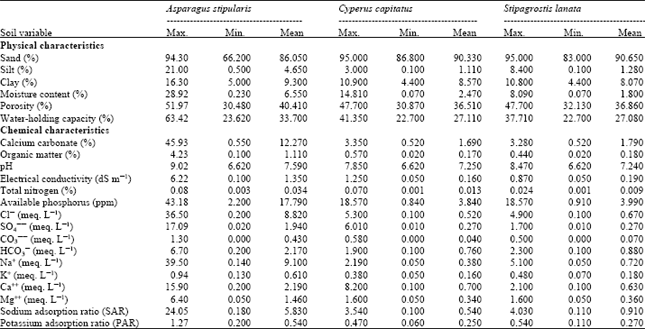
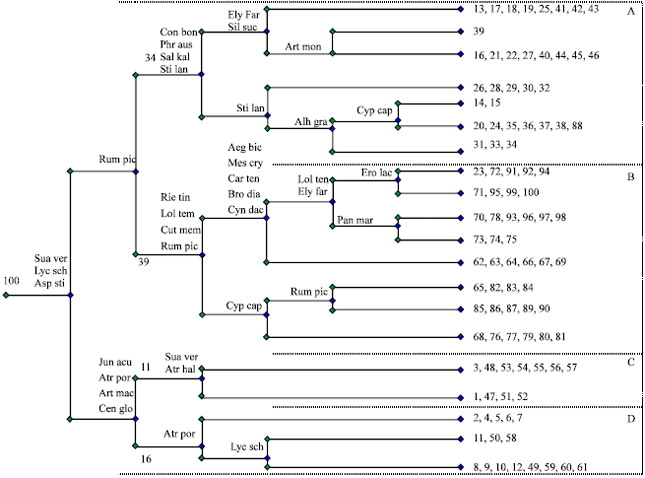
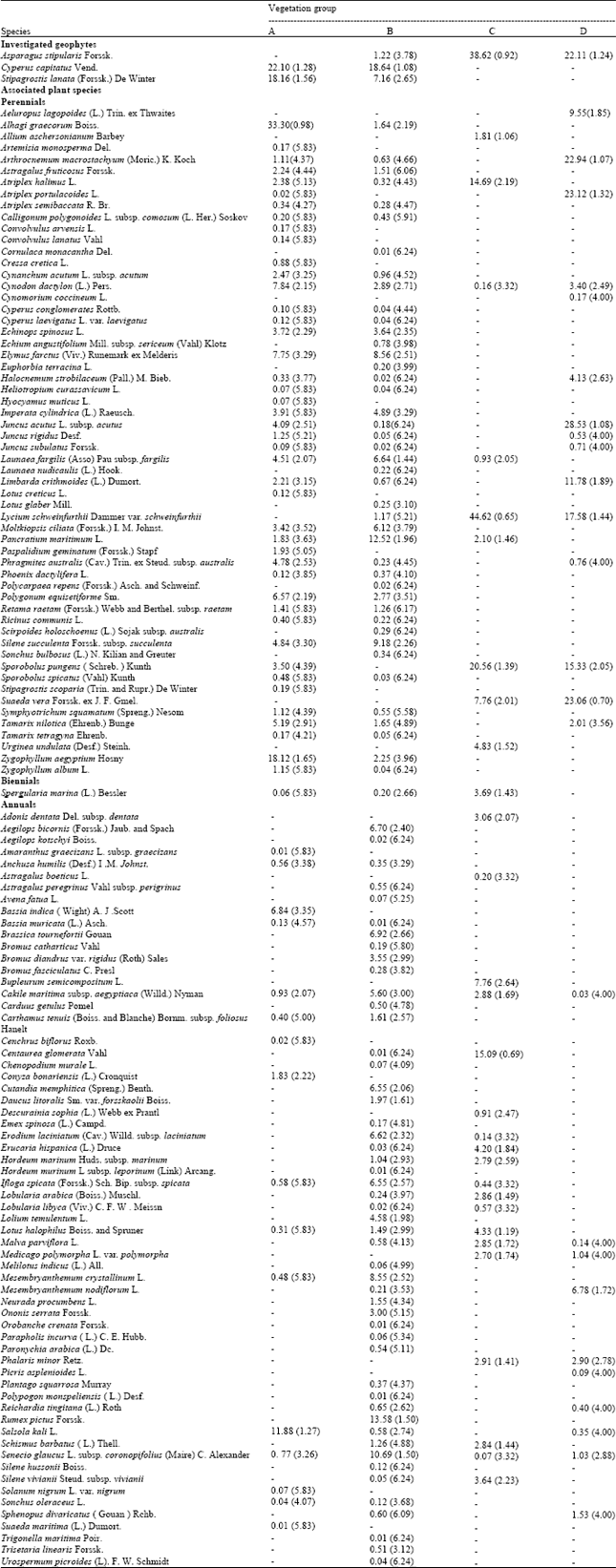
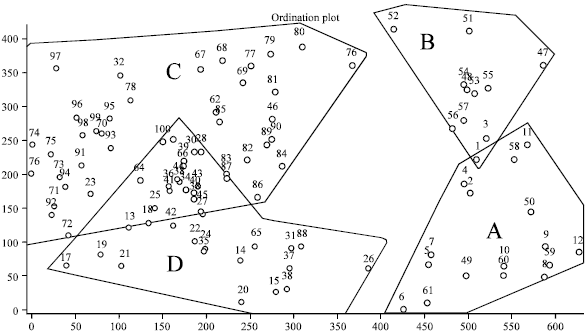
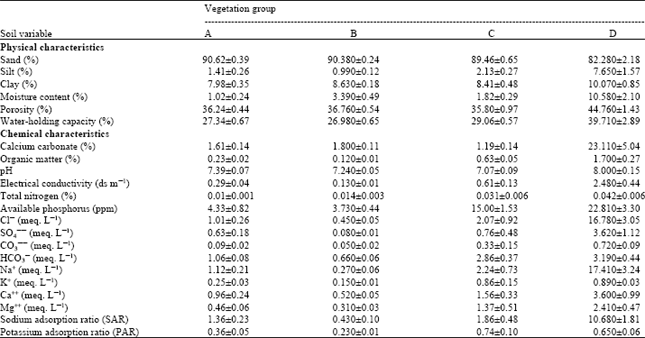
Research Article
Ecological Investigation of Three Geophytes in the Deltaic Mediterranean Coast of Egypt
Department of Agricultural Botany, Faculty of Agriculture, Tanta University, Tanta, Egypt
Abdelnaser A. Elzaawely
Department of Agricultural Botany, Faculty of Agriculture, Tanta University, Tanta, Egypt