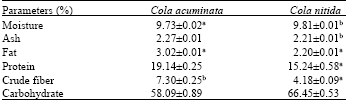
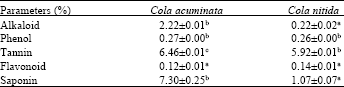
Research Article
Proximate and Phytochemical of Cola nitida and Cola acuminata
Departmen t of Chemical Sciences, Afe-Babalola University, Ado-Ekiti, Nigeria
D.F.A. Dewumi
Departmen t of Chemical Sciences, Afe-Babalola University, Ado-Ekiti, Nigeria
J.Y.T. Alabi
Department of Biological Sciences, Afe-Babalola University, Ado-Ekiti, Nigeria
A. Adegoke
Department of Chemistry, Federal University of Technology, Akure, Nigeria