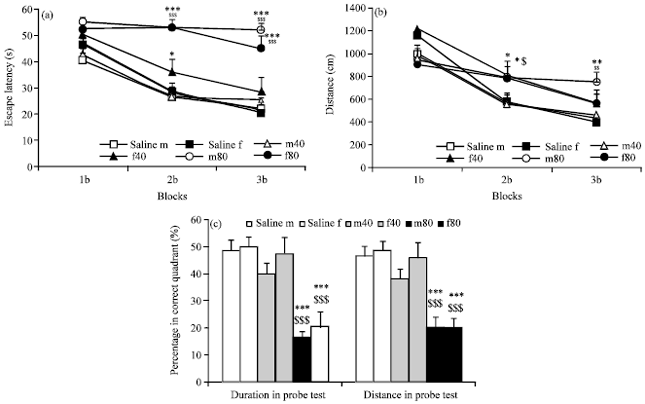
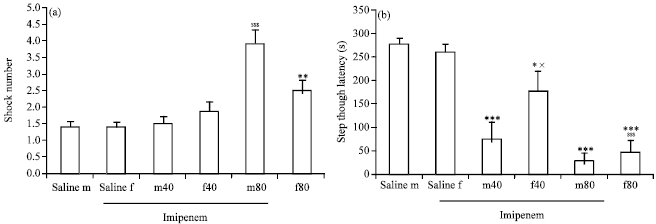
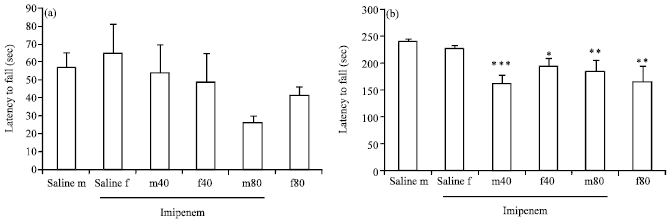
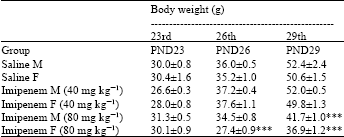
Research Article
Hippocampus and Cerebellum Function Following Imipenem Treatment in Male and Female Rats: Evaluation of Sex Differences During Developmental Stage
Physiology Research Center, Kerman University of Medical Sciences, Kerman, Iran
Lale Golchin
Physiology Research Center, Kerman University of Medical Sciences, Kerman, Iran
Ali Asghar Vahidi
Pediatrics Department, Kerman University of Medical Sciences, Kerman, Iran
Mohammad Shabani
Neuroscience Research Center, Kerman University of Medical Sciences, Kerman, Iran