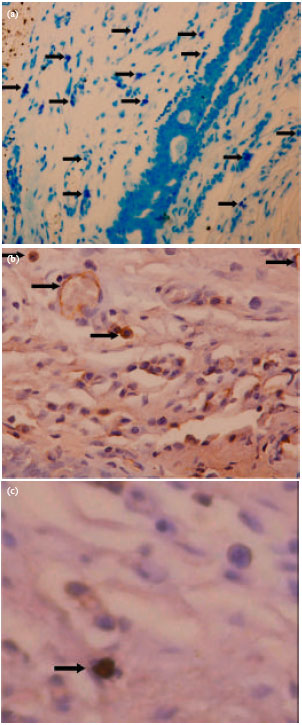
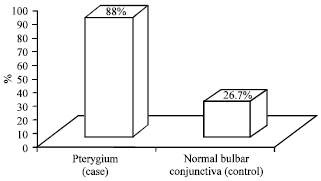
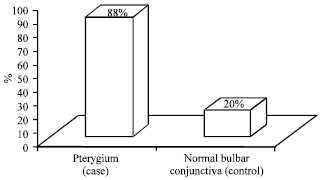
Research Article
Concomitant Examination of Inflammation and Angiogenesis in the Pathogenesis of Primary Moderate Pterygium in a Well-designed Case-control Study
Department of Pathology, Tabriz University of Medical Sciences, Sina Hospital, Tabriz, Iran
Abdollah Shenasi
Department of Ophthalmology, Tabriz University of Medical Sciences, Nikookari Hospital, Tabriz, Iran