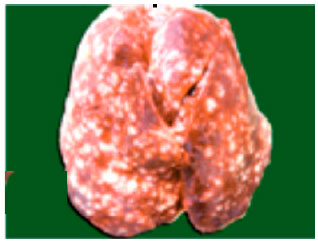
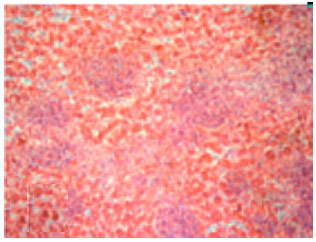
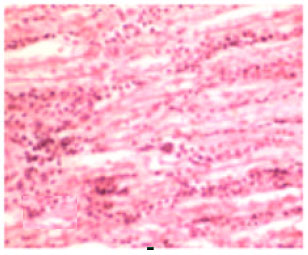
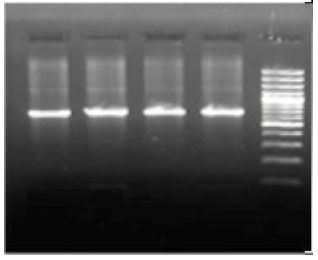
Review Article
Recent Trends in Diagnosis and Control of Marek’s Disease (MD) in Poultry
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (U.P.)-243 122, India
Rajamani Barathidasan
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (U.P.)-243 122, India
Asok Kumar
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (U.P.)-243 122, India
Rajib Deb
Molecular Genetics Laboratory Animal Genetics and Breeding, Project Directorate on Cattle, Indian Council of Agricultural Research, Grass farm Road, Meerut, Uttar Pradesh-250001
Amit Kumar Verma
Department of Veterinary Epidemiology and Preventive Medicine, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Vishvidhyalaya Ewam, Go-Anusandhan Sansthan (DUVASU), Mathura (U.P.)-281001, India
Kuldeep Dhama
Avian Diseases Section, Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (U.P.)-243 122, India