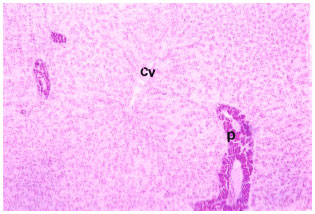
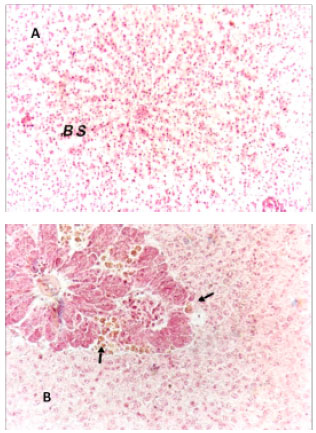
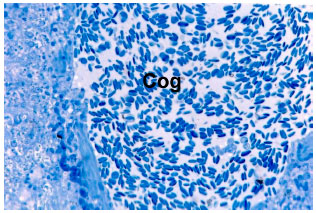
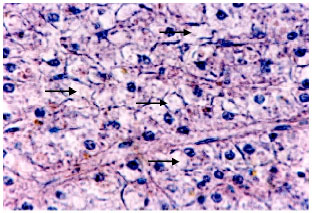
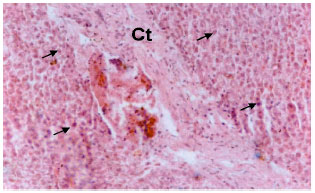
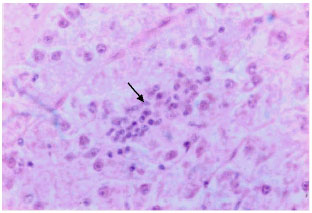
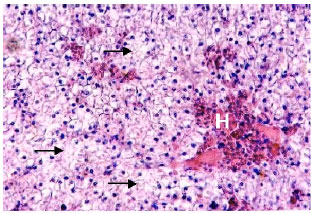
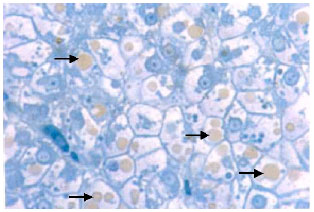
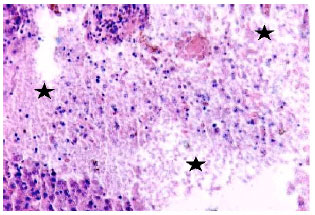
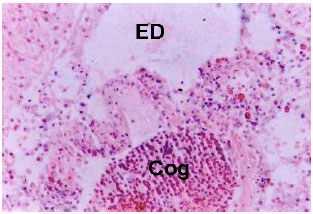
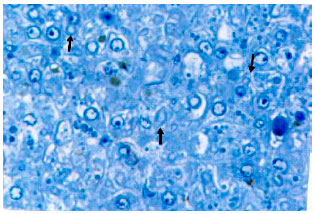
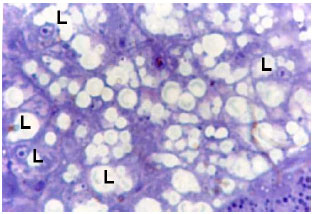
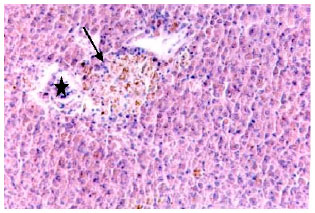
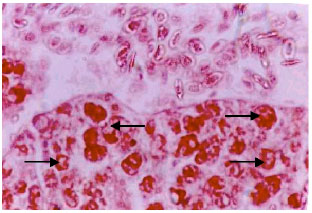
Research Article
Effect of Lead Nitrate on the Liver of the Cichlid Fish (Oreochromis niloticus): A Light Microscope Study
Department of Zoology, Faculty of Science, Assiut University, Egypt
Imam A.A. Mekkawy
Department of Biology, Faculty of Science, Taif University, Saudi Arabia
Ahmed S.A. Harabawy
Department of Biology, Faculty of Science, North Jeddah, King Abdul Aziz University, Saudi Arabia
Abdel Salam M.I. Ohaida
Department of Zoology, Faculty of Science, 7 October Misurata University, Libya