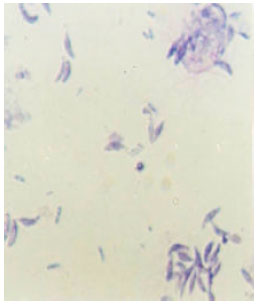
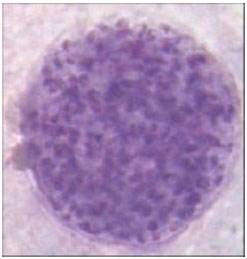
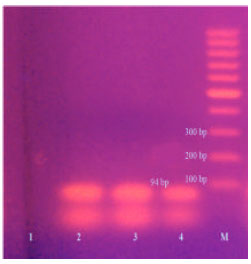
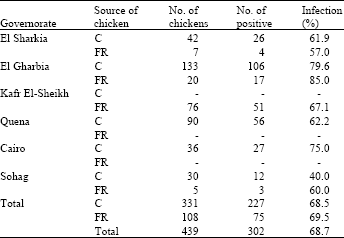
Research Article
Zoonotic Chicken Toxoplasmosis in Some Egyptians Governorates
Division of Veterinary Research, Department of Zoonotic Diseases, National Research Center, Giza, Egypt
Lobna Mohamed Ali Salem
Department of Zoonotic Diseases, Faculty of Veterinary Medicine, Benha University, Egypt
Adel M. Abdel-Aziz El-Newishy
Department of Zoonotic Diseases, Faculty of Veterinary Medicine, Benha University, Egypt
Raafat Mohamed Shaapan
Division of Veterinary Research, Department of Zoonotic Diseases, National Research Center, Giza, Egypt
Ehab Kotb El-Mahllawy
Department of Zoonotic Diseases, Faculty of Veterinary Medicine, Sohag University, Egypt