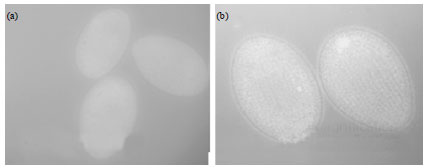
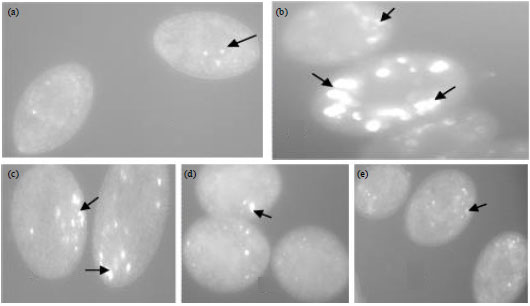
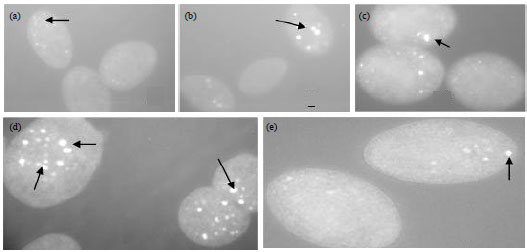
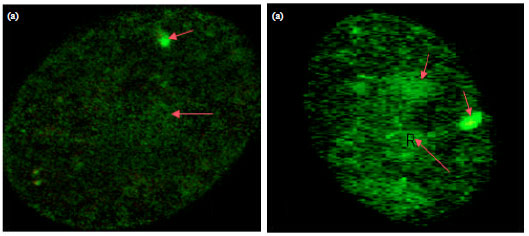
Research Article
Identification of Dopamine Receptor in Tetrahymena thermophila by Fluorescent Ligands
Helmholtz Zentrum M�nchen, German Research Center for Environmental Health, Institute of Ecological Chemistry, Ingolst�dter Landstrasse 1, D-85764 Neuherberg, Germany
Gerd Pfister
Helmholtz Zentrum M�nchen, German Research Center for Environmental Health, Institute of Ecological Chemistry, Ingolst�dter Landstrasse 1, D-85764 Neuherberg, Germany
Karl-Werner Schramm
Helmholtz Zentrum M�nchen, German Research Center for Environmental Health, Institute of Ecological Chemistry, Ingolst�dter Landstrasse 1, D-85764 Neuherberg, Germany