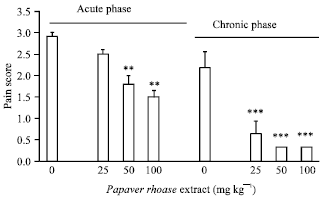
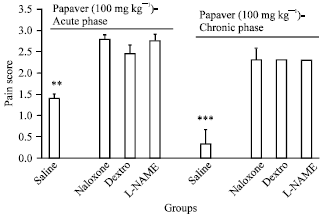
Research Article
Effects of Papaver rhoeas (L.) Extract on Formalin-induced Pain and Inflammation in Mice
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
M. Ranjbaran
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
F. Jafari
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
A. Najafi-Abedi
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
B. Rahmani
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
B. Esfandiari
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
B. Delfan
Razi Herbal Medicine Research Center, Lorestan University of Medical Sciences, Khoramabad, Iran
N. Mojabi
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
M. Ghahramani
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran
H. Sahraei
Neuroscience Research Center, Baqyiatallah (A.S.) University of Medical Sciences, Tehran, Iran