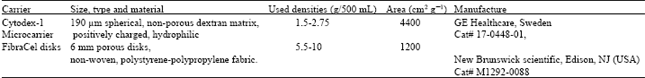
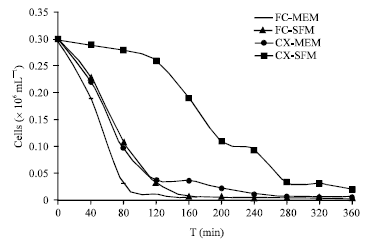
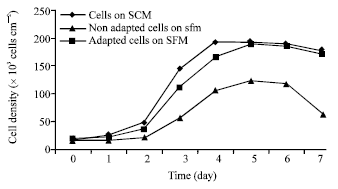
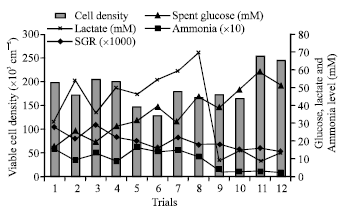
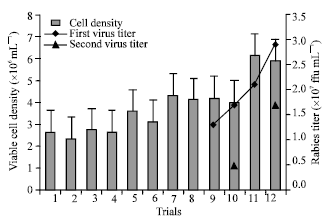
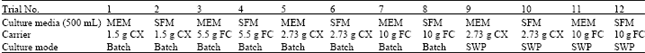Research Article
High Vero Cell Density and Rabies Virus Proliferation on Fibracel Disks Versus Cytodex-1 in Spinner Flask
WHO-Collaborating Centre for Reference and Research on Rabies, Pasteur Institute of Iran, Tehran, Iran
Ali Zavareh
Department of Viral Vaccine Production, Pasteur Institute of Iran
M. Ali Shokrgozar
National Cell Bank of Iran, Pasteur Institute of Iran
Ali Ramezani
Department of Biotechnology, School of Pharmacy, Zanjan University of Medical Sciences, Zanjan, Iran
Ahmad Fayaz
WHO-Collaborating Centre for Reference and Research on Rabies, Pasteur Institute of Iran, Tehran, Iran