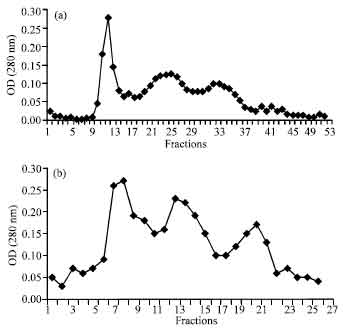
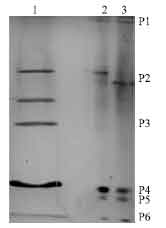
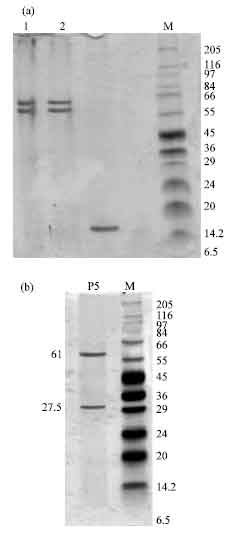
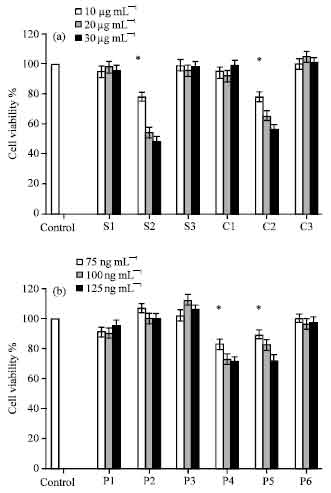
Research Article
Immunosuppressive Proteins Isolated from Spiral and Coccoid Cytoplasmic Solutions of Helicobacter pylori
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
A. Akbarzadeh
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
A. Farhangi
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
M. Mehrabi
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
Z. Hosseinian
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
M. J. Mokhtari
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran
Z. Saffari
Department of Pilot Biotechnology, Pasteur institute of Iran, Tehran, Iran