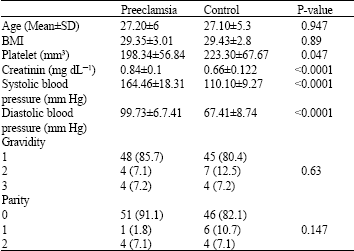
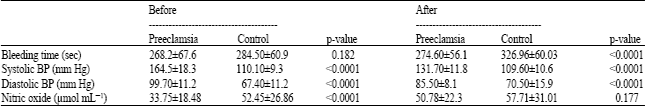
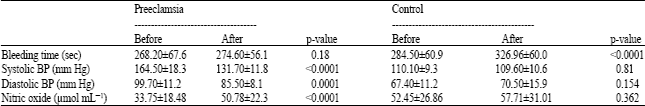
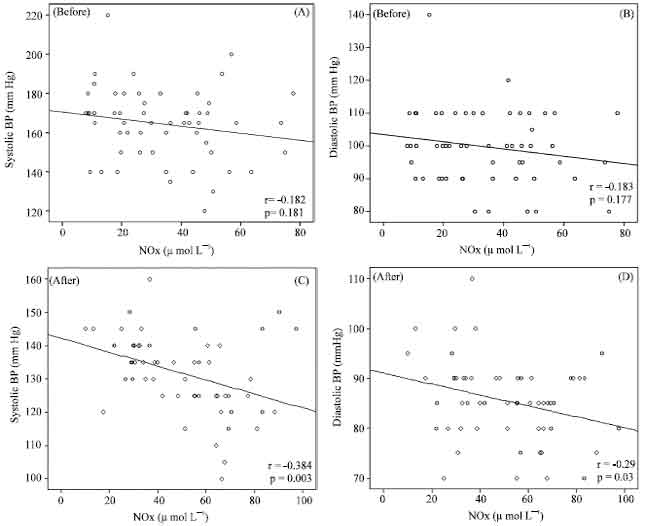
Research Article
The Effect of Magnesium Sulfate on Bleeding Time and Nitric Oxide Production in Preeclamsia
Department of Gynecology and Obstetrics, Imam Khomeini Teaching Hospital, Mazandaran University of Medical Sciences, Sari, Iran
Alireza Rafiei
Molecular Cell Biology Research Center, Mazandaran University of Medical Sciences, Sari, Iran
Fereshteh Yazdani
Department of Gynecology and Obstetrics, Imam Khomeini Teaching Hospital, Mazandaran University of Medical Sciences, Sari, Iran
Zahra Hosseini-khah
Molecular Cell Biology Research Center, Mazandaran University of Medical Sciences, Sari, Iran
Keyvan Yusefnezhad
Department of Cardiology, Fatameh Zahra Hospital, Mazandaran University of Medical Sciences, Sari, Iran