Research Article
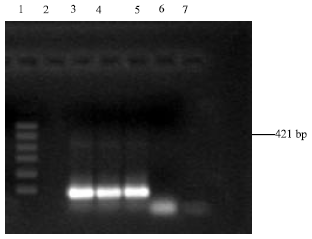
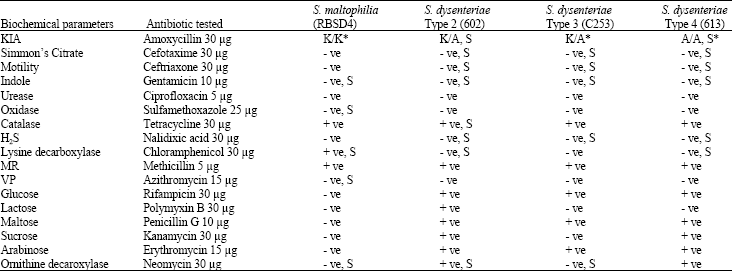
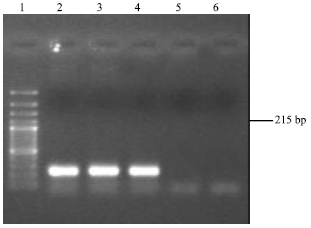
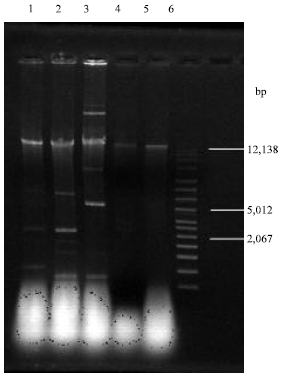
Virulence of Environmental Stenotrophomonas maltophilia Serologically Cross-reacting with Shigella-specific Antisera
Department of Microbiology, University of Dhaka, Dhaka-1000, Bangladesh
N. Azmuda
Department of Microbiology, University of Dhaka, Dhaka-1000, Bangladesh
S.I. Khan
Department of Microbiology, University of Dhaka, Dhaka-1000, Bangladesh
N.K. Birkeland
Department of Biology, University of Bergen, P.O. Box 7803, N-5020, Bergen, Norway
M.Z. Rahman
Virology Laboratory, Laboratory Science Division, International Centre for Diarrhoeal Diseases Research, P.O. Box 128, Dhaka-1000, Bangladesh