ABSTRACT
The purpose of this study is to examine the effects of neurosecretory material (NSM) in Corpus Allatum (CA) of endoparasitic Pimpla turionellae L. on reproduction. For this purpose, the egg maturation of the insect and the amount of the NSM in CA of the serial section of the brain were studied. Egg maturation was determined by measuring the terminal oocyte length. In order to determine the amount of the NSM in CA the neurosecretory granules of which were stained by paraldehyde fuchsin are considered. The egg maturation of P. turionellae have performed twice. The NSM intensity in CA continued increasingly from the first day of egg development until the first oviposition days; and reached the maximum level before the second egg maturation phase. These observations showed that the NSM in CA of this insect may be related to egg development.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2010.748.752
URL: https://scialert.net/abstract/?doi=pjbs.2010.748.752
INTRODUCTION
The median neurosecretory cells of the brain were shown as the control center of reproduction (Toyoda et al., 1999) and other metabolic activities (Shimokawa et al., 2008; Broughton et al., 2010). These cells in different orders were studied during the reproduction period of female insect (Juberthie and Caussanel, 1980). Formerly, it was specified that the brain cells which are stained purple by paraldehyde fuchsin techniques include neurosecretory material (Peric-Mataruga et al., 2001). There are many researches on these neurosecretory cells which are stained purple by paraldehyde fuchsin (Toyoda et al., 1999). These cells were searched concerning egg maturation on Labidura riparia (Juberthie and Caussanel, 1980) and Lymantria dispar (Leob and Hayes, 1980). The fact that, the NSM which is produced in the median neurosecretory cells of the brain reaches to the corpora allata by passing through the corpora cardiaca via the nervi corporis cardiaci is well known. In CA of some insects, the NSM which is stainable with paraldehyde fuchsin positively, was presented (Sakiko, 2003). In insects, corpora allata are known to be involved in metabolic activities (Weaver and Edwards, 1990; King et al., 2005; Tombes and Smith, 2005). Although, some authors demonstrated the presence of the NSM within the CA only a few of them provided some detailed account of the functional significance of the NSM in the CA. In some studies it was mentioned that CA also has correlation with egg maturation (Toyoda et al., 1999; Huerta and Martinez, 2008).
As seen above, the relationship between the neuroendocrine system of the insects belonging to different orders and the reproduction activities were studied and various results were obtained. Previously the head endocrine system of P.turionellae had been given morphologically, histologically (Ozluk, 1991) and ultrastructurally but the physiological relationships between the endocrine system and the reproduction activity had not been mentioned. On the insects merged to Hymenopter order, it is rare to encounter studies that show the correlation between the brain neurosecretory cells and the reproduction (Ozluk, 1993b). Especially, there is no study about the reproduction period which is related to the correlation between the NSM in CA and the egg development of endoparasitic P. turionellae.
In this study, the variation of the NSM intensity in CA during the reproduction period of female P. turionellae was observed. Thus, it was aimed to enlighten the relationship between the neurosecretory material in corpora allata and reproduction in P. turionellae.
MATERIALS AND METHODS
Experimental animals: Individuals of female P. turionellae was reared under laboratory conditions between years 2003-2005 in the Insect Culture Laboratuary at Ankara University, Faculty of Science, Department of Biology. The female experimental animals were obtained from these stock culture.
P. turionellae was reared at the Experimental Animals Laboratuary of the Ankara University Biology Department between 2003-2005 years. The female experimental animals were obtained from the stock culture. The continuity of the stock culture was supplied from the greater wax moth, Galleria mellonellae, reared in the semi-synthetic diet (Bronskill, 1961). All P. turionellae females were kept at temperature of 25±2°C with relative humidity of 75±5% in a 12: 12 (L: D) photoperiod. They were fed with cotton pieces absorbed with 50% honey solution and to each insect was given a pupa of G. mellonellae every two days in order to satisfy their host haemolymph needs. All the experimental virgin females were reared without males to eliminate possible effects of mating (Pratt and Davey, 1972b; Acle et al., 1990). They were selected on the 0, 3, 6, 9, 12, 15, 18, 21, 24 and 27th days, after adult emergence. All experimental females were selected in the same body size to eliminate the difference in terminal oocyte length (Briegel, 1990).
Microscopy: All females were decapitated at the same time (14.00-17.00) to eliminate the possible diurnal changes in the neurosecretory system (Engelmann, 1968). The dissected brain of P. turionellae were fixed in Bouin’s solution for 12-18 h, sectioned serially at 7 milimicron after being embedded in paraffin and were stained with paraldehyde fuchsin after potassium permanganate oxidation (Gomori, 1950). The rest of the body of the decapitated animals was dropped into isotonic insect saline and was left for no longer than 3 h. The ovaries were dissected out, for microscopic calculation, under a binocular microscope and were fixed in Bouin’s fixative 4 h. Dissected ovaries were sectioned serially at 10 milimicron. The sections were stained with Ehrlich’s haemotoxylin and eosin.
Measurements: In order to point out the changes in the intensity of NSM on the experiment days, the paraldehyde fuchsin positive granules in CA were counted. The NSM in the gland was numbered 0 to 5 increasingly according to its staining intensity (Adams et al., 1975). An average value for each day investigated was obtained by dividing the sum of the staining intensity of each CA into the number of CA of each the day. These calculations were done for each insect separately.
The lengths of the terminal oocyte were measured with an ocular micrometer. The developmental stages of the oocyte growth were recorded as the length of the biggest terminal oocyte in ovariole (Pratt and Davey, 1972a). All measurements were made in the largest area in the cross sections of the terminal oocytes. Only one biggest terminal oocyte length from each of at least 10 animals in each studied day was measured and their averages were recorded (Table 1).
| Table 1: | The NSM intensity of CA and the terminal oocyte length change of P. turionellae on different maturation days |
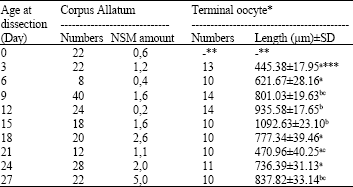 | |
*Only one terminal oocyte were measured in each female; **There is no terminal oocyte in ovariole on the day of emergence; ***Means compared vertically. Means not followed by the same superscript are significantly different from each other at p<0.05 by Fisher’s t-test | |
The measurements in the terminal oocytes were done by Fisher’s (1948) method of significance control test (p<0.05) between two means.
RESULTS
Terminal oocyte growth: The larger oocytes in any ovariole represent a longitudinally arranged series in which the terminal oocyte nearest the base of the ovariole is the oldest and largest oocyte representing successively earlier stages in the oocyte maturation (Pratt and Davey, 1972a). In all P. turionellae females, there is no differentiation in the ovariole on the day of emergence (Table 1). The terminal oocytes continue to increase in length between the 3rd day and 15th day. Between the 12th day and 15th day, the length of the terminal oocyte reaches to maximum level which is called primary egg maturation stage. Between the days 15th and 21th, the length of the terminal oocyte decreases to minimum. These days on which the terminal oocyte length decreased to minimum are called the first oviposition stage. The terminal oocyte again continues to increase after the 21th day on the stage of secondary egg maturation.
Neurosecretory materials in corpus allatum: The NSM intensity in corpus allatum had different levels during the reproductive period of the females of P. turionellae (Table 1). The NSM intensity of newly emerged insects was on a limited scale and the level was low. This intensity level increased on the first days depending on the oocyte maturation, showed fluctuation on the following days. The NSM intensity which decreased the minimum level before the first egg laying phase days, reached a high intensity on the first egg laying days. After the first egg laying days, the NSM amount decreased considerably, but on the second egg maturation phase, it reached to the maximum level. This level of the NSM intensity in CA during egg maturation can be mentioned as there is a correlation between the CA and egg maturation on the reproduction period in this insect.
DISCUSSION
A brain cell has been considered as neurosecretory if stained purple with paraldehyde fuchsin and only the purple condition has been considered indicative of the presence of NSM and has been called paraldehyde fuchsin positive. The fact that the CA of insects includes NSM was shown by the studies done by means of light and electron microscope (King et al., 2005; Tombes and Smith, 2005; Bonetti et al., 2006). In the mean time the NSM exists also in the CA of female of P. turionellae was marked in the serial sections which were obtained from the head of the insect. In many hitherto studies on insects, the neurosecretory cells have appeared to regulate oocyte growth (Greenplate et al., 1985) by activating the CA (Bhaskaran and Jones, 1980; Khan et al., 1984; Ulrich et al., 1985) to produce a hormone essential for protein transfer from haemolymph into the developing oocytes for yolk deposition (Bell and Barth, 1971) and perhaps by controlling protein synthesis and metabolism themselves (Osorio et al., 1997). In many studies, the brain hormones have already been chosen to show their cyclic activity (Leob and Hayes, 1980) correlated with the oocyte maturation (Tabakoglu, 1988; Toyoda et al., 1999). Cyclical changes in the appearance and volume of the CA have been observed in many insects. The volume has often been used as a measure of the gland activity (Osorio et al., 1997; Huerta and Martinez, 2008). Just as, the CA volume has been used as a criterion to show the cyclic changes in the terminal oocyte maturation (Stay and Tobe, 1981; Ulrich et al., 1985). In one of his studies on P. turionellae, Ozluk (1993a) indicated a parallelism between the volume of the gland and its NSM amount.
In this study, in P. turionellae females, the day of emergence was characterized by the lack of differentiation in the ovariole. On this day, the amount of the NSM in CA was at a certain level. During the first days of the terminal oocyte growth, a permanent increase or decrease was not observed at the amount of the NSM in CA. It was suggest that after the first days of egg maturation, this level showed fluctuation. Indeed, the level which has the minimum value just before the first egg laying days (on 12th day), increased considerably on the first egg laying days (between 18th day and 21st day). This reduction which occurred just before terminal oocytes reached to the maximum length was assessed as there has been no more need to hormones since the egg maturation was completed. It has been known that the NSM in brain passes to CA throughout corpora cardiaca. Thus, the changes in the intensity of NSM in CA can be explained with the changes in the intensity of NSM in brain. It was specified that the NSM which is secreted from median neurosecretory cells, provides oocyte maturing (Adams et al., 1968). They specified with another researcher group in 1975 that there is a feedback mechanism between the brain cells of M. domestica and ovariums. Adams (1976) also specified that the oocytes which complete the maturation, produce a hormone which blocks the secretion activity of brain that is called oostatic hormone; and this hormone causes NSM accumulation in brain on egg laying phase. I also found that the striking increase in NSM of P. turionellae on the first egg laying days can be explained with the suggestion of Siew (1965) which shows that egg laying activity requires high level of hormone. When injected the brain extract which is obtained from the egg laying females of Gryllus bimaculatus to young mature females, Sefiani (1987) observed that they also show egg laying activity and abdominal contractions starts. Thus, it was recommended that more neurohormones are required for egg laying and this neurohormone causes abdominal contractions and egg laying movements. After the first egg laying phase, the NSM in CA of P. turionellae decreases seriously. The NSM amount which decreases after the first egg laying phase of P. turionellae, reached to its maximum level on the second egg maturation days. In fact that the NSM intensity which has a considerably high level during the egg laying phase, decreased after the egg laying activity completed in L. riparia (Juberthie and Caussanel, 1980), in 9-24 h after the egg laying activity in Galeruca tanaceti (Siew, 1965) and in 12-36 h in M. domestica (Adams et al., 1975). The present study suggests that this decrease which occurred at the beginning of the second egg maturation might have occurred for the same reasons as the first decrease in P. turionellae. The maximum increase in the corpus allatum activity at the height of their gonadotrophic activity has been described by a considerable number of authors and my findings are consistent with those of given by Stay and Tobe (1981), Pratt and Pener (1983). In this study this maximum increase may be true for P. turionellae only in the second oocyte maturation phase, because its maximum increase occurred after the 21th day. Dia and Gilbert (1993) proposed that the largest volume of the CA was reached at the beginning of yolk storage in the terminal oocytes. Also, it was purposed that the CA is especially responsible for the second oocyte growth in Nauphoeta cinerae (Buschor et al., 1984).
The results obtained from present study shows that the NSM amount in corpus allatum of P. turionellae changes during the reproductive activity. It is also estimated that the egg laying and the egg maturation phases of P. turionellae are controlled by corpus allatum hormones could be denoted from these results.
REFERENCES
- Acle, D., V.J. Brookes, G.E. Pratt and F.R. Rene, 1990. Activity of the corpora allata of adult female Leucophaea maderae: Effects of mating and feeding. Arch. Insect Biochem. Physiol., 14: 121-129.
Direct Link - Adams, T.S., A.M. Hintz and J.G. Pomonis, 1968. Oostatic hormone production in houseflies, Musca domestica, with developing ovaries. J. Insect Physiol., 14: 983-993.
CrossRef - Adams, T.S., S. Grugel, P.I. Ittycherian, G. Olstad and J.M. Caldwell, 1975. Interactions of the ring gland, ovaries and juve-nile hormone with brain neurosecretory cells in Musca domestica. J. Insect Physiol., 21: 1027-1043.
CrossRef - Adams, T.S., 1976. ovaries, ring gland, after neurosecretion dur-ing the second gonotrophic cycle in the housefly, Musca domestica. Gen. Comp. Endocrinol., 30: 69-76.
CrossRef - Osorio, S., M.D. Piulachs and X. Belles, 1997. Feeding and activation of corpora allata in the cockroach Blattella germanica (L.) (Dictyoptera, Blattellidae). J. Insect Physiol., 44: 31-38.
CrossRef - Bonetti, A.M., A.C. Siquieroli, F.A. Santana, C.U. Vieira and M. Bezerra et al., 2006. Effect of juvenile hormone III on the ultrastructure of the Corpora allata in Melipona quadrifasciata (Hymenoptera, Apidae, Meliponını). Braz. J. Morphol. Sci., 23: 393-400.
Direct Link - Briegel, H., 1990. Metabolic relationship between female body size, reserves and fecundity of Aedes aegypti. J. Insect Physiol., 36: 165-172.
CrossRefDirect Link - Buschor, J., P. Beyeler and B. Lanzrein, 1984. Factors re-sponsible for the initiation of a second oocyte maturation cycle in the ovoviviparous cockroach Nauphoeta cinerae. J. Insect Physiol., 30: 241-249.
CrossRef - Dia, J. and I.L. Gilbert, 1993. An ultrastructural and developmental analysis of the corpus allatum of juvenile hormone deficient mutants of Drosophila melanogaster. Dev. Genes Evol., 202: 85-94.
Direct Link - Greenplate, J.T., R.L. Glaser and H.H. Hagedorn, 1985. The role of factors from the head in the regulation of egg development in the mosquito Aedes aegypti. J. Insect Physiol., 31: 323-329.
CrossRef - Huerta, C. and M.I. Martinez, 2008. Morphological changes in reproductive organs and neuroendocrine centers related to nesting, mating and larvicide behavior in Eurysternus mexicanus harold (Scarabaeinae: Eurysternini). Coleopterists Bull., 62: 123-132.
Direct Link - Juberthie, C. and C. Caussanel, 1980. Release of brain neurosecretory products from the neurohaemal part of the aorta during egg-laying and egg-care in Labidura riparia, (Insecta: Dermaptera). J. Insect Physiol., 26: 427-429.
CrossRef - Khan, M.A., H.M. Romberg-Privee and H. Schooneveld, 1984. Innervation of the corpus allatum in the Colorado potato beetle as revealed by retrograde diffusion with horseradish peroxidase. Gen. Comp. Endocrinol., 55: 66-73.
Direct Link - King, R.C., S.K. Aggarwal and D. Bodenstein, 2005. The comparative submicroscopic cytology of the corpus allatum-corpus cardiacum complex of wild type and fes adult female Drosophila melanogaster. J. Exp. Zool., 161: 151-175.
Direct Link - Loeb, M.J. and D.K. Hayes, 1980. Neurosecretion during dia-pause and diapause development in brains of mature embryos of the gyp-sy moth, Laymantria dispar. Ann. Entomol. Soc. Am., 73: 432-436.
Direct Link - Pratt, G.E. and K.G. Davey, 1972. The corpus allatum and oo-genesis in Rhodnius prolixus (Stal.): I. The effects of allatectomy. J. Exp. Biol., 56: 215-222.
Direct Link - Pratt, G.E. and K.G. Davey, 1972. The corpus allatum and oo-genesis in Rhodnius prolixus (Stal.): III. The effect of mating. J. Exp. Biol., 56: 223-237.
Direct Link - Pratt, G.E. and M.P. Pener, 1983. Procecene sensitivity of cor-pora allata in adult female Locusta migratoria after electrocoagulation of the pars intercerebralis neurosecretory cells. J. Insect Physiol., 29: 33-39.
CrossRef - Sefiani, M., 1987. Regulation of egg laying and in vitro oviducal contractions in Gryllus bimaculatus. J. Insect Physiol., 3: 215-222.
CrossRef - Siew, Y.S., 1965. The endocrine control of adult reproductive diapause in the chrysomelid beetle, Galeruca tanaceti (L.) III. J. Insect Physiol., 11: 973-981.
CrossRef - Stay, B. and S.S. Tobe, 1981. Control of the corpora allata during a reproductive cycle in a viviparous cockroach. Am. Zool., 21: 663-674.
Direct Link - Tombes, A. and D.S. Smith, 2005. Ultrastructural studies on the corpora cardiaca-allata complex of the adult alfalfa weevil, Hypera postica. J. Morphol., 132: 137-147.
Direct Link - Toyoda, I., H. Numata and S. Sakiko, 1999. Role of the median neurosecretory cells in the ovarian development of the blow fly Protophormia terraenovae. Zool. Sci., 16: 187-199.
Direct Link - Weaver, R. and J.P. Edwards, 1990. The role of the corpora allata and associated nerves in the regulation of ovarian cycles in the oviparous cockroach Periplaneta Americana. J. Insect Physiol., 36: 51-59.
CrossRef - Broughton, S.J., S.J. Slack , C. Alic, N. Metaxakis and A. Bass et al., 2010. DILP-producing median neurosecretory cells in the Drosophila brain mediate the response of lifespan to nutrition. J. Anat., 9: 336-346.
CrossRef - Sakiko, S., 2003. Anatomy and functions of brain neurosecretory cells in diptera. Microsc. Res. Tech., 62: 114-131.
PubMed








