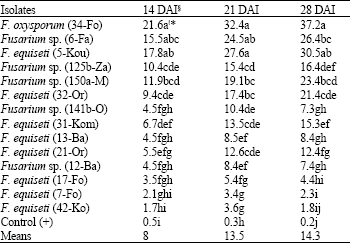
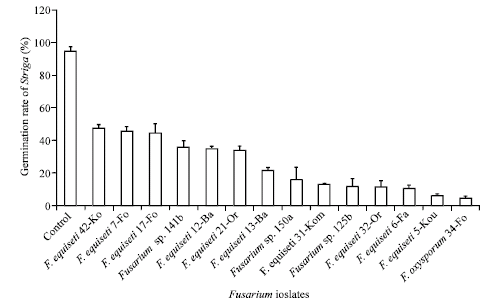
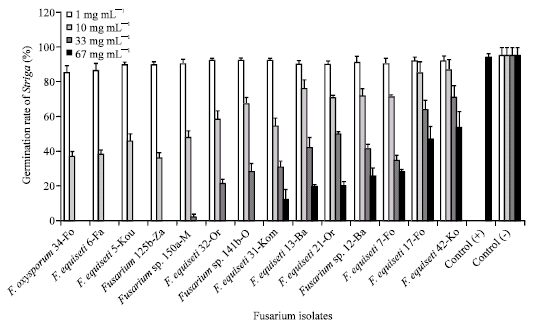
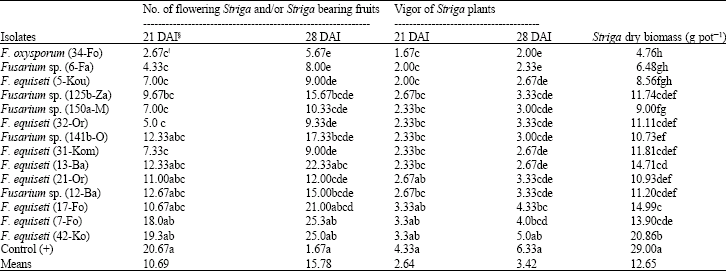
Research Article
Pathogenicity of Fusarium isolates to Striga hermonthica in Burkina Faso
Institut de l�Environnement et de Recherches Agricoles, 04 B.P. 8645 Ouagadougou 04, Burkina Faso
H. Traore
Institut de l�Environnement et de Recherches Agricoles, 04 B.P. 8645 Ouagadougou 04, Burkina Faso
P. Sereme
Institut de l�Environnement et de Recherches Agricoles, 04 B.P. 8645 Ouagadougou 04, Burkina Faso
D. E. Hess
Merry Lea Environmental Center of Goshen College, Wolf Lake, IN, USA
P. Sankara
Universite de Ouagadougou, Unite de Formation et de Recherche en Sciences de la vie et de la terre, B.P. 7021, Ouagadougou, Burkina Faso