Research Article
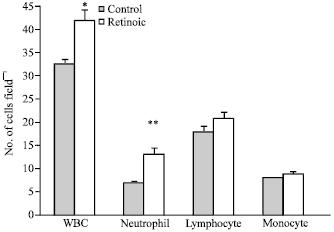
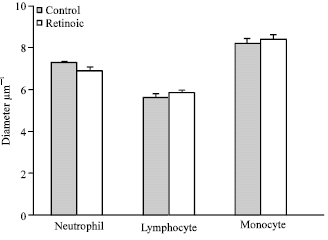
The Effects of All-Trans Retinoic Acid on Leukocytes in Rat's Embryo
Department of Anatomical Sciences, Semnan University of Medical Sciences, Semnan, Iran
H. R. Samene
Department of Anatomical Sciences, Semnan University of Medical Sciences, Semnan, Iran