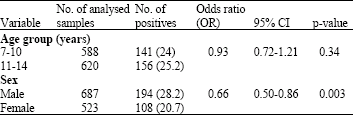
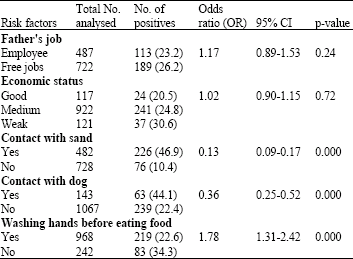
Research Article
Seroprevalence of Toxocariasis in Schoolchildren in Northern Iran
Department of Parasitology and Mycology
A. Daryani
Department of Parasitology and Mycology
G. Barzegar
Department of Parasitology and Mycology
M. Nasrolahei
Department of Microbiology and Immunology
A. Khalilian
Department of Biostatistics, Sari Medical School, Mazandaran University of Medical Sciences, Sari, Iran