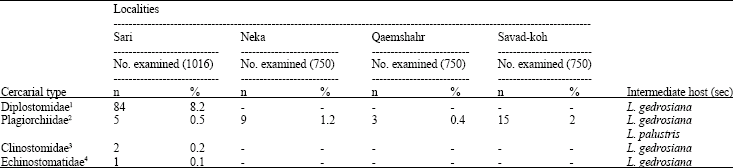
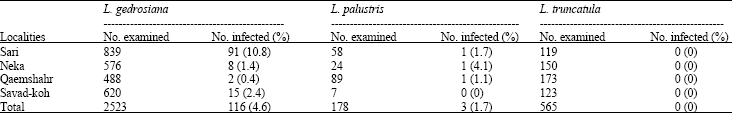
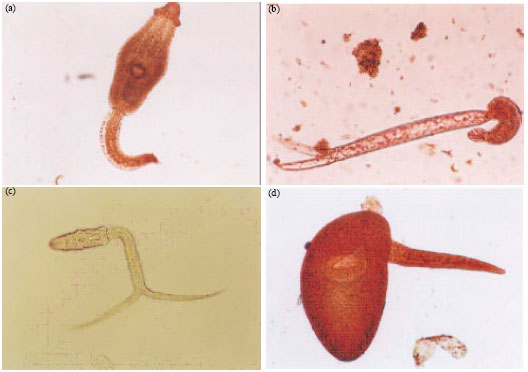
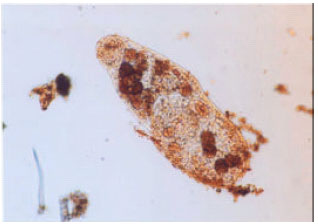
Research Article
A Faunistic Survey of Cercariae Isolated from Lymnaeid Snails in Central Areas of Mazandaran, Iran
Department of Parasitology and Mycology, Mazandaran University of Medical Sciences, Sari, Iran
A. Daryani
Department of Parasitology and Mycology, Mazandaran University of Medical Sciences, Sari, Iran
S. A. Karimi
Department of Parasitology and Mycology, Mazandaran University of Medical Sciences, Sari, Iran