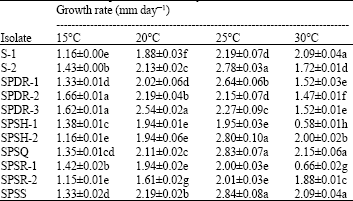
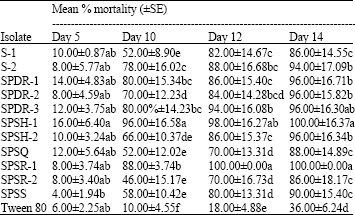
Research Article
Beauveria bassiana Characterization and Efficacy vs. Sunn Pest, Eurygaster integriceps Puton (Hemiptera: Scutelleridae)
General Commission for Scientific Agricultural Research, Aleppo Center, P.O. Box 4195, Aleppo, Syria
M. El-Bouhssini
ICARDA, P.O. Box 5466, Aleppo, Syria
M. Jamal
Faculty of Agriculture, Damascus University, Damascus, Syria
A. N. Trissi
Faculty of Agriculture, Aleppo University, Aleppo, Syria
Z. Sayyadi
ICARDA, P.O. Box 5466, Aleppo, Syria
M. Skinner
Entomology Research Laboratory, University of Vermont, Burlington, Vermont, USA 05405-0105
B. L. Parker
Entomology Research Laboratory, University of Vermont, Burlington, Vermont, USA 05405-0105