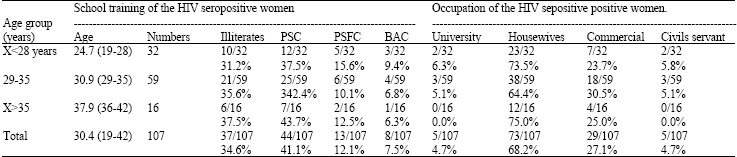
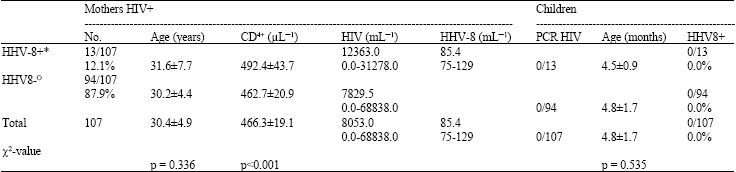
Case Study
Mother-to-Child HIV and HHV-8 Transmission in Neonates at Saint Camille Medical Centre in Burkina Faso
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
J. Simpore
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
D.S. Sanou
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
D. Karou
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
D.J. Sia
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
D. Ouermi
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
C. Bisseye
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
T. Sagna
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
S. Odolini
University of Brescia, Piazza Spedali Civili, 1-25123 Brescia, Italy
F. Buelli
University of Brescia, Piazza Spedali Civili, 1-25123 Brescia, Italy
V. Pietra
Biomolecular Research Center Pietro Annigoni, CERBA, 01 BP 364 Ouagadougou 01, Burkina Faso
S. Pignatelli
Biomolecular Research Center Pietro Annigoni, CERBA, 01 BP 364 Ouagadougou 01, Burkina Faso
C. Gnoula
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
J.B. Nikiema
University of Ouagadougou, 07 BP 5252 Ouagadougou, Burkina Faso
F. Castelli
University of Brescia, Piazza Spedali Civili, 1-25123 Brescia, Italy