Research Article
Hepatitis C Infection and Apoptosis in Hepatocellular Carcinoma
Department of Biology, Teacher College, King Saud University, P.O. Box 271323, Riyadh 11352, Saudi Arabia
Hepato-Cellular Carcinoma (HCC) ranks among the 10 most common cancers worldwide. It evolves from several chronic liver diseases, most of which culminate in cirrhosis. As the most common causes, other than alcoholic cirrhosis, are chronic hepatitis B and C infections, its prevalence worldwide is linked to the prevalence of these two viruses (Seeff and Hoofnagle, 2006). Since, the discovery of Hepatitis C Virus (HCV) in 1989, there has been much debate regarding its potential modes of transmission particularly as only about half of the reported cases of acute HCV have a defined parenteral exposure. It has been clearly established that blood-blood contact is important in its producing conflicting evidence (Clarke and Kulasegaram, 2006).
Hepatitis C Virus (HCV) has a high propensity to persist and to cause chronic hepatitis C, eventually leading to cirrhosis. Since, HCV itself is not cytopathic, liver damage in chronic hepatitis C is commonly attributed to immune-mediated mechanisms. Cytotoxic lymphocytes kill HCV-infected cells via the perforin/granzyme pathway, but also release Fas ligand and inflammatory cytokines such as interferon gamma (IFN-γ). Release of soluble effectors molecules helps to control HCV infection, but may also destroy uninfected liver cells and can attract further lymphocytes without HCV specific inflammatory cells will expand the tissue damage triggered by HCV infection and ultimately activate fibrogenesis (Spengler and Nattermann, 2007).
Cytokines and chemokines are proteins that play a critical role in the regulation of immunity and inflammation in patients with chronic hepatitis C. Thus, Cytokines, chemokines and apoptosis level reflect the progression of inflammation and fibrosis in hepatitis C infected patients but their signatures differ (Neuman et al., 2007).
Apoptosis can be induced by many physiological and pathophysiological stimuli, including specific receptor molecules. Such as CD95, Tumor Necrosis Factor (TNF) receptor, but also by many stress forms, such as growth factor withdrawal, irradiation, heat shock, bacteria, toxins and viruses (Smith et al., 1994). Apoptosis is a key mechanism underlying fulminate hepatic failure. Apoptosis in the liver is generated mainly by Fas system (Pelli et al., 2007). The host immune response to HCV infection is composed of both a non-specific immune response, including interferon (IFN) production and Natural Killer (NK) cell activity and a virus-specific immune response, including humoral and cellular components. Susceptibility to infection has been related to immunological disturbances (Lotfy et al., 2006).
Fas (APO-1,CD95) is a 45 KDa cell surface glycoprotein that belongs to the Tumor Necrosis Factor (TNF)/Nerve Growth Factor (NGF) receptor family. It transducers apoptotic signals to the cell when agonistic anti-Fas antibody or Fas ligand (Fas-L) binds with it (Iio et al., 1998). Apoptosis receptors, especially Fas, are widely expressed in all liver cell types, likely in response to the evolutionary pressure to eliminate hepatotropic viruses. The Fas/Fas-L system is indeed the pathway most commonly used by immunocytes to kill virally infected cells (Walczak and Krammer, 2000).
In chronic hepatitis C, the expression of Fas antigen on hepatocytes is upregulated and Fas ligand expression is detected on liver infiltrating mononuclear cells. Thus, Fas antigen/Fas Ligand-mediated apoptosis is thought to be involved in hepatic injury in chronic hepatitis C (Iio et al., 1998; Ali et al., 2003).
So, the present study aimed to evaluate the Fas/Fas-L levels in each of non-cancerous individual (controls), HCC and non-HCC patients.
All of the experimental procedures were conducted in Department of Biological and Medical Research, King Faisal Specialist Hospital and Research Centre, Riyadh, Saudi Arabia between 2007 and 2008. Forty cancerous patients and Non-cancerous individuals (Controls). Twenty controls were recruited from blood bank. Ten milliliters of venous blood were withdrawn from all patients and controls, the blood was left to coagulate following by centrifugation, serum and buffy coat layer were separated by Ficoll solution and aliquoted according to (Ferrante and Thong, 1980).
Separation of buffy coat layer: Five milliliters of peripheral blood was put in a vacutainer tube containing heparin (50 U mL-1 blood) and gently mixed. The blood was then dilutes with 4 mL of PBS in a 15 mL clean sterile tube and mixed. After centrifugation at 1800 rpm for 20 min at room temperature, a clear visible layer of buffy coat was formed at the interface between plasma and Ficoll-Hypaque. The buffy coat layer was aspirated using a pasteur pipette and the cell pellet was resuspended in 10 mL PBS, mixed well, centrifuged at 1800 rpm for 10 min then the supernatant was discarded. The cells were resuspended once more with PBS and the previous step was repeated twice at 1500 and 900 rpm 10 min each for the second and third washes, respectively. The buffy coat were counted using hemocytometer. Their number was adjusted to 3000-5000 cell mL-1 using PBS. Cell viability was determined using trypan blue exclusion test. Cells are then preserved in Demso and RPMI solution in a 10% suspension to keep them viable and put in -80°C until used.
All samples were tested for Hepatitis C Virus Antibodies (HCV-Ab) by solid phase enzyme linked immonosorbent assay (ELISA), alpha-fetoprotein (AFP) by AxSYM which was based on the Microparticle Enzyme Immuno Assay (MEIA) technology for quantitative determination (Abbott Laboratories, USA).
Detection of HCV-Ab: Test kit was a solid phase enzyme linked immonosorbent assay (ELISA), for the detection of the antibodies to HCV in human serum ADALTIS ITALIA SPA). Two hundred microliters of sample diluent was added to well and 200 μL of the negative control, positive control and the samples as diluted (1:10 with sample diluent) then added into respective wells. Microplate was incubated at 37°C for 60 min followed by 5 times washing Buffer. One hundred microliters of conjugate solution was added to all wells followed by incubating at room temperature for 30 min followed by washing. 100 μL of the substrate solution was added to all wells and then incubated at room temperature for 30 min. 100 μL of stop solution was added to all wells. The color intensity of the solution in each well was measured by using microplate reader at 450 nm.
Alpha-fetoprotein (AFP): Sample, specimen diluent and Anti-AFP coated microparticles were delivered to one well of the reaction vessel. During the incubation of this reaction mixture, the AFP in the specimen binds to the Anti-AFP coated microparticles forming an antibody-antigen complex. An aliquot of the reaction mixture was transferred to the Matrix cell. The microparticles bind irreversibly to the glass fiber matrix. The Matrix cell was washed to remove unbound materials. The Anti-AFP: Alkaline phosphatase conjugate was dispensed onto the Matrix cell and binds to the antibody- antigen complex. The Matrix cell was washed to remove unbound materials. The substrate, 4-methylumbelliferyl phosphate, was added to the matrix cell and the fluorescent product was measured by the MEIA optical assembly.
RNA extraction: RNA extracted by RNA isolation kit by Gentra system, USA. Two hundred microliters of Buffy coat added to 1 mL of cell lysis solution, proper mixing was done by inverting tube several times to lyse the cells. The supernatant containing the RNA was transferred to a clean sterile eppindorf and added to it 1 mL 100% isopropanol and left into an ice bath overnight Mixing was done by inverting the sample gently 50 times. Then centrifugation at 1500 rpm for 5 min, Supernatant was discarded and added to the RNA 1 mL of 70% ethanol, proper mixing by inverting several times to wash RNA. Centrifugation for 2 min at 1500 rpm then ethanol was carefully discarded off. Then was left to dry in thermo block for about 1 h at 56°C. Thirty microliter of RNA-hydration solution was added and mixed well then stored until RT-PCR was done for detection Fas/Fas-L.
HCV-RNA extraction from serum: Hepatitis C virus-RNA was extracted from serum by isolation kit (Gentra system, USA) according to method described by (Boom et al., 1990). Lysis of cells and viruses took place and released Nucleic Acid (NA) was bound to the NA carrier, forming complexes which could be rapidly sedimented by centrifugation. Two hundred microliters of serum was pipetted into a reaction vessel containing 900 μL of GuSCN containing lysis buffer and 40 μL of solid NA carrier (size fractionated silica particles) then mixed well followed by incubating at room temperature for 10 min and centrifuged at 13000 rpm for 15 sec. The supernatant was aspirated and pellet was washed twice with GuSCN containing washing buffer by adding 1 mL each time, vortex and centrifugation followed by aspiration of the liquid. The pellet was then washed twice with 70% ethanol and once with acetone. After removing the supernatant, the tube was incubated at 56°C incubator with its lit opened for 10 min until completely dry. Sixty microliters of nuclease free water was added for elution of NA. The tubes were then centrifuged at 13000 rpm for 15 min and supernatant was carefully collected and centrifuged again for another 15 min at 13000 rpm and was used for the RT step.
Reverse transcription: The reverse transcribed into cDNA amplified by PCR and then Fas receptor Fas (APO-1,CD95) cell surface glycoprotein that belongs to the Tumor Necrosis Factor (TNF) detected and determination of Fas Ligand (Fas-L), which of confirmation of apoptotic activity by Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR). About 12.5 μL of extracted RNA was used, to which 12.5 μL of RT mixture was added (10 mM Tris-HCl (pH 8.8), 50 mM KCl, 0.1% Triton X-100, 6 mM MgCl2, 20 U of RNase inhibitor, 0.6 mM (each) deoxynucleotide triphosphate (dNTPs)-Sigma-25 ng of HCV-6 primer (5’ACC TCC3’), 200 U superscripte RTI (Gibco-BRL, Gaitherburg, MD).
Polymerase chain reaction: About 2.5 μL Taq polymerase (Perkin-Elmer Cetus), 50 mM Tris-HCl (pH 8.3), 1.5 mM MgCl2, 0.2 mM (each) dNTPs (Sigma), 100 ng RB-6A (Sense primer 5’GTG AGG AACTACTGTCTTCACG 3’, 100 ng RB-6B (Antisense primer 5’ ACT CGC AAG CACCCTATCAGG3’[nt 292 to 312]), 12.5 μL of the cDNA.
RT-PCR: The RT-PCR products were detected by 2% agarose gel electrophoresis in 1xTBE buffer. The gel was stained with 5 μL of 10 mg mL-1 ethidium bromide. After the gel was completely set, the comb were carefully removed and the gel was mounted in the electrophoresis tank filled with 1xTBE buffer. Ten microliters of the PCR product was mixed with 3 μL gel loading buffer and slowly loaded into the slot of the submerged gel. Also, a 100 bp DNA marker (Gibco-BRL) was loaded along as a size marker. One hundred and seventy five voltage for 25 min was then applied and the gel was examined under the ultraviolet and photographed. A positive result revealed a PCR product band at 265 bp using a size marker of the Φ X 174/Hae III DNA (Williams, 1998).
Aminotransferases (ALT/AST), Alanine-aminotransferase (ALT/GPT) and AST/GOT activities were determined by a kinetic method according to (Klin, 1970). Kits supplied by Centronic GmbH-Germany.
Biostatistics: The data obtained in this study were statistically analyzed with using the t-test and Z-test.
The data show that the AST, ALT, AFP values in positive and negative HCV-RNA of HCC patients appear markedly increasing compared with controls and thus they are recording highly significant (p<0.05) difference (Table 1).
Comparison of Fas-expression for HCV-Ab, HCV-RNA between controls and each of HCC, non HCC. In positive HCV-Ab, we found that elevation of Fas- expression in HCC and in non- HCC is insignificant higher than in normal donors. In negative HCV-Ab, Fas expression in HCC is highly significant (p<0.05) but in non-HCC is insignificant. In positive HCV-RNA, Fas-receptors in HCC and in non-HCC patients are insignificant increase than in normal donors.
In negative HCV-RNA, Fas-expression in HCC patients is significant elevation (p<0.05) than the normal donors, but in non-HCC is insignificant. The comparison of Fas-L expression for HCV-Ab and HCV- RNA between normal donors and each of HCC and non-HCC is shown in Table 2. In positive HCV-Ab, Fas-L expression in HCC patients is insignificant by comparing with controls, whereas it is significant (p<0.05) in non-HCC. In negative HCV-Ab, Fas-L in HCC patients and in non-HCC patients is insignificant. In positive HCV-RNA, Fas-L in HCC and non-HCC patients is insignificant. In negative HCV-RNA, Fas-L in HCC and in non-HCC patient is insignificant (Table 2).
From above data, we observed that incidence of down-regulation of Fas-L with the viral C infection especially in case of controls. There is a good negative correlation between Fas-L expression and presence of viral C infection in HCC and non-HCC patients.
| Table 1: | Biochemical Comparison parameters between groups in relation to HCV |
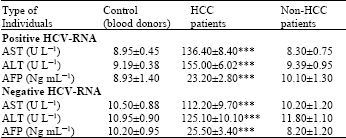 | |
| Data are expressed as Mean±SE. ***Highly significant at p<0.05 | |
| Table 2: | Comparison of Fas-expression and Fas-L expression between groups in relation to HCVAb and HCV-RNA |
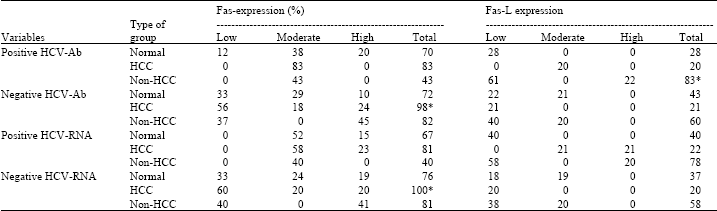 | |
| *Significant at p<0.05 | |
The study showed significant elevation of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in each of positive and negative viral hepatitis C in HCC patients only. The reasons for elevated aminotransferases are unreliable only the attack of viral infection but on some co-factors (steatosis- immunosuppressed state) associated with progression of fibrosis and development of cirrhosis (incidence of hepatocellular carcinoma). These results were in agreement with Tarao et al. (1999), Ayman et al. (2006) and Moriyama et al. (2004), who reported that the incidence of HCC differed according to the ALT levels in chronic HCV patients and liver cirrhosis patients.
The reason for AFP elevation may be according to (Li et al., 2005), who reported that AFP is able to promote the expression of Fas-L and TRAIL (tumor necrosis factor related apoptosis-inducing ligand) in hepatoma cells and enhance the expression of Fas and TRAILR in lymphocytes. These could elicit the escape of hepatocellular carcinoma from the host’s lymphocytes immune surveillance.
Wong and Lee (2006) showed that it is more likely that HCV causes HCC through an intermediate step of cirrhosis. Chronic HCV infection causes mild chronic inflammation of the liver. On going cycles of inflammation, necrosis and apoptosis eventually lead to scarring (fibrosis) and ultimately, severe bridging fibrosis with nodular regeneration (cirrhosis).
This study showed that Fas-upregulations don’t change in case of positive viral C infection related to HCC and in non-HCC as well. In contrast in negative HCV-RNA, Fas-expression in HCC patients is significant.
These results are disagreement with (Noriyasu et al., 2000), which demonstrated that a close relationship exists between hepatitis C Virus (HCV) infection of Peripheral Blood Mononuclear Cells (PBMCs) and cell-surface Fas expression in patients with hepatitis C and showed the possibility of PBMCs apoptosis via a Fas-mediated system. In addition, the treatment of patients’ PBMCs with anti-Fas antibody induced cell death, with nuclear additional stimulation such as soluble Fas-L might induce apoptosis in these Fas-expressing cells.
Abnormalities of biochemical parameters are sometimes indirectly correlated with the Fas-expression. These results proved that to some extent there is a direct correlation between Fas and disease development. This result is in agreement with (Kondo et al., 1997), who reported that the killing of the hepatocytes could be blocked by soluble Fas-L protein, suggesting the importance of Fas-activation in the disease development.
There are also several studies agree with our findings and the conclusions of the present study (Higuchi and Gores, 2003), who reported that death receptor mediated cell death mechanism has also been implicated in liver injury in cholestasis and alcoholic liver disease.
In present study, the incidence of down-regulation of Fas-L with viral-C infection occurs. The cause for minimizing of Fas-L expression may be according to Iken et al. (2006), who reported that enhanced apoptosis of activated lymphocytes was associated with up-regulation of Fas-L by HCV transgenic hepatocytes and was specifically inhibited by anti-Fas-L blocking antibody. In brief, the Fas-upregulations don’t change in case of positive viral C infection related to HCC and also in non-HCC. But in negative HCV-RNA, Fas-expression in HCC patients is significant.
In this study, we can conclude that neither Fas nor Fas-L may be affected by viral C infection but Fas is actually influenced by the type of disease like HCC. These findings are consistent with Higuchi and Gores (2003), who reported that in cholestasis-induced hepatocyte death, Fas is required, but Fas-L is not.