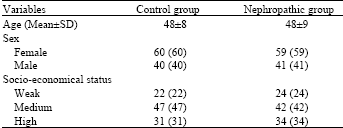
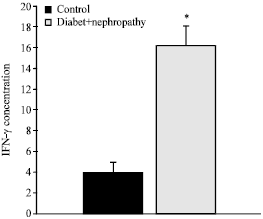
Research Article
Evaluation of IFN-γ Serum Level in Nephropatic Type 2 Diabetic Patients
Department of Laboratory Sciences, Faculty of Medicine, Islamic Azad University, Zahedan Branch, Zahedan, Iran
M.K. Arababadi
Department of Microbiology, Hematology and Immunology, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
G. Hassanshahi
Department of Microbiology, Hematology and Immunology, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
N. Yaghini
Department of Biochemistry, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
V. Pooladvand
Department of Biochemistry, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
Ali Shamsizadeh
Department of Phisiology, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
E.R. Zarandi
Department of Microbiology, Hematology and Immunology, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran
H. Hakimi
Department of Microbiology, Hematology and Immunology, Faculty of Medicine, Rafsanjan University of Medical Science, Rafsanjan, Iran