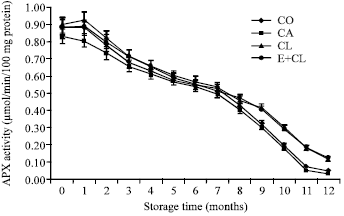
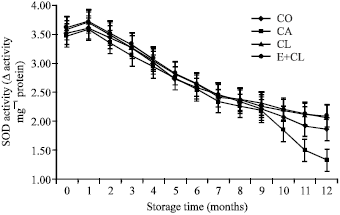
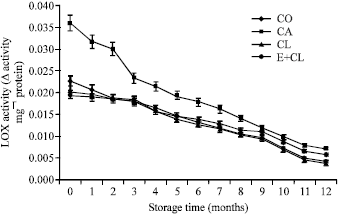
Research Article
The Perspective Effects of Various Seed Coating Substances on Rice Seed Variety Khao Dawk Mali 105 Storability II: The Case Study of Chemical and Biochemical Properties
Department of Agricultural Technology, Faculty of Technology, Mahasarakham University, Talard Sub-District, Muang District, Maha Sarakham 44000, Thailand
W. Pan-in
Plant Variety Protection Research Group, Plant Variety Protection Division, Department of Agriculture, Ministry of Agriculture and Cooperatives, Royal Thai Government, 10900 Bangkok, Thailand
E. Pawelzik
Section of Quality of Plant Products, Institute of Agricultural Chemistry, Georg-August University of Goettingen, Carl-Sprengel-Weg , D-37075, Goettingen, Germany
S. Vearasilp
Department of Agronomy, Faculty of Agriculture, Chiang Mai University, 50000, Thailand