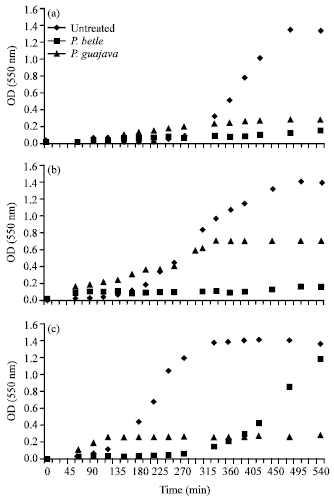
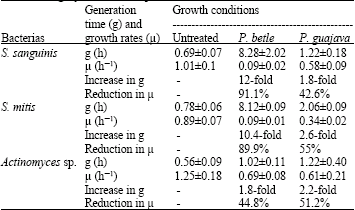
Research Article
Bacteriostatic Effect of Piper betle and Psidium guajava Extracts on Dental Plaque Bacteria
Department of Oral Biology, Faculty of Dentistry, University of Malaya, 50603 Kuala Lumpur, Malaysia
Z.H.A. Rahim
Department of Oral Biology, Faculty of Dentistry, University of Malaya, 50603 Kuala Lumpur, Malaysia
Y. Othman
Institute of Biological Sciences, Faculty of Science, University of Malaya, 50603 Kuala Lumpur, Malaysia
M. Yusoff
Institute of Biological Sciences, Faculty of Science, University of Malaya, 50603 Kuala Lumpur, Malaysia