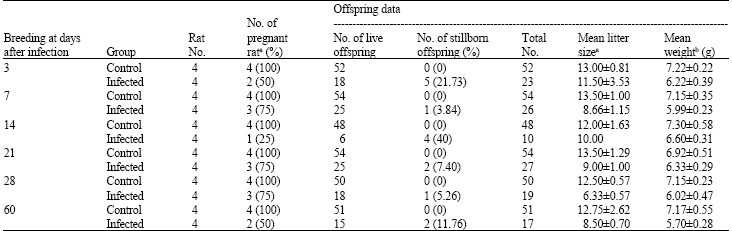
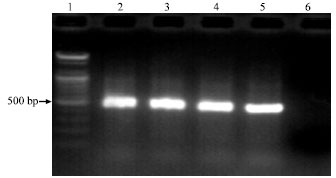
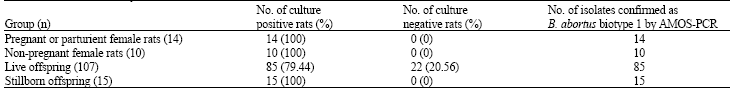Research Article
Effects of Brucella abortus Biotype 1 Infection on the Reproductive Performance of Sprague-Dawley Rats
College of Veterinary Medicine, Chonbuk National University, Jeonju 561-756, Republic of Korea
Mst. Minara Khatun
College of Veterinary Medicine, Chonbuk National University, Jeonju 561-756, Republic of Korea
Byeong Kirl Baek
College of Veterinary Medicine, Chonbuk National University, Jeonju 561-756, Republic of Korea
Sung Il Lee
Division of Model Animal, Institute of Biomedical Science, Kansai Medical University, Fumizono 10-15, Moriguchi, Osaka, 570-8506, Japan