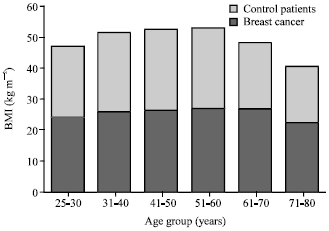
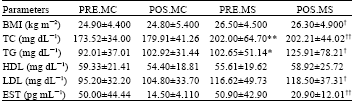
Research Article
Serum Lipid Profile of Breast Cancer Patients
Department of Molecular Medicine, School of Medical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
S. Donkor
Department of Molecular Medicine, School of Medical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
B. Wiafe Addai
Peace and Love Clinic, Oduom, Kumasi, Ghana
N. Amidu
Department of Molecular Medicine, School of Medical Sciences, College of Health Sciences, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana









ajil Abed Al-Hussein Reply
Effect of Lipid brofile on breast cancer , and also effect life style on breast cancer
Dr.phyo min min oo Reply
relationship between lipid profile and breast cancer?
Vibrators Reply
The results of this study suggest that increased serum lipid profiles may associate with breast cancer risk in Thai women. Further studies to group important factors including, cancer stages, types of cancer, parity, and menopausal status that may affect to lipid profiles in breast cancer patients along with an investigation of new lipid profiles to clarify most lipid factors that may involve in breast cancer development are needed.