Research Article
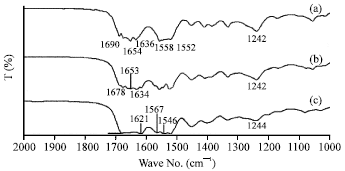
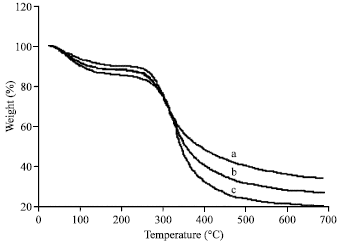
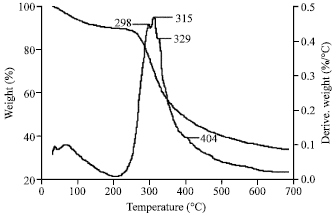
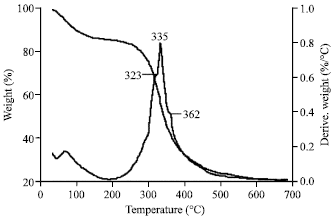
Morphology, Secondary Structure and Thermal Properties of Silk Fibroin/Gelatin Blend Film
Division of Secretary, Department of Chemistry,
Srisuwan Yaowalak
Division of Secretary, Department of Chemistry, Faculty of Science, Mahasarakham University, 44150, Thailand
Simcheur Wilaiwan
The Central Instrument, Faculty of Science, Mahasarakham University, 44150, Thailand
Srihanam Prasong
Center of Excellence for Innovation in Chemistry,3Center of Excellence for Innovation in Chemistry, Department of Chemistry, Faculty of Science, Mahasarakham University, 44150, Thailand