Research Article
A Diet Rich in Leafy Vegetable Fiber Improves Cholesterol Metabolism in High-Cholesterol Fed Rats
Department of Food Science and Nutrition, National Research Center, P.O. Box 12311, Giza, Egypt
Traditional Mediterranean food, which usually consists of vegetables, fruits, spices, pasta, olive oil, seafood and only a little red meat, has been termed Mediterranean diet (Hadjichambis et al., 2008). The consumption of fruits and vegetables has often been correlated with lower rates of coronary heart disease, diabetes and cancer and with greater longevity (Thrichopoulou et al., 2000; Goulet et al., 2003), not only in Mediterranean, but also in non-Western populations of India (Maffi, 2001). The healthy effects of fiber, especially its cholesterol-lowering properties, have been ascribed to the moiety of soluble dietary fiber (Aller et al., 2004).
Jew’s mallow (Corchorus olitorius) is a dark green leafy vegetable, high in protein, Dietary Fiber (DF), calcium, iron and β-carotene. It is used in Egypt, Cyprus and Tunisia, as well as in Southern Asia, Jordan, West Africa, Malaysia, Philippines and Central America (Hadjichambis et al., 2008; Innami et al., 2005; Oboh et al., 2009; Oboh and Akindahunsi, 2004; Zeghichi et al., 2003). In Egypt, it is consumed as a traditional dish called Molokheya and in rural villages many families grow their own Jew’s mallow as it constitutes a significant component of their diet (Hadjichambis et al., 2008; Shrif et al., 1980).
The awareness of the healthy effects of Jew's mallow Molokheya is not very high. By identifying culturally accepted foods of a high nutrient quality and thereby combating human diseases, the general heath of the community can be improved. The objective of the present work was to assess-in vivo some nutritional properties derived from regular consumption of leaf vegetable (Jew's mallow), especially its hypocholesterolemic effect in high-cholesterol fed rats. As Jew’s mallow contains about 90% water, it was freeze-dried in order to concentrate its micronutrients in the diet.
Samples preparation: Fresh samples of Jew’s mallow were collected from the two main markets in Egypt, 6 October and Al-Obour. The purchase of the Jew’s mallow was made by experimenters’ assistants, who were instructed to use the same considerations as an ordinary consumer would do, evaluating quality with price. They immediately sent to the National Research Center, the Jew’s mallow purchased, in a manner suitable to retain its freshness. If any portions did not arrive fresh, they were discarded. In the laboratory, the fresh Jew’s mallow was washed, inedible portions (62%) were removed and only edible parts (leaves) were used. The Jew’s mallow leaves samples were composited into one.
The dish of Jew’s mallow was prepared following traditional standard Egyptian procedures. The basic recipe is to add 400 g of finely chopped fresh Jew’s mallow leaves to two cups of chicken broth or water in a vessel that was placed over a low fire. In the meantime, the Jew’s mallow spice mix was prepared by frying a tablespoon of ground-dried coriander (2.5 g) and three cloves of minced garlic (3 g) in a teaspoon of olive oil in a separate pan until they turned golden brown. Just before the Jew’s mallow and the broth or water started boiling, the spice mix was added to the vessel and immediately removed from the fire.
| Table 1: | Composition of freeze dried Jew’s Mallow per 100 g edible portion (% dry matter) |
 | |
| Values are means of triplicate analysis±SEM | |
It is very important not to allow the Jew’s mallow to boil as it coagulates and becomes inedible. The resulting Jew’s mallow dish is either consumed as a soup, or served with white rice and boiled- or grilled chicken. In the present study, we did not add the Jew’s mallow spice mix to be sure that the observed physiological effect originated from Jews mallow plant not from other constituents of dish (coriander, garlic, olive oil). In addition, we used water instead of chicken broth.
The Jew’s mallow dish (without Jew’s mallow spice mix, prepared by water) was prepared for analysis and experiments with the animals by homogenization in a blender and weighed for freeze-drying. The freeze-dried samples were finely ground and the weighed samples were taken for the determination of moisture. The weights of the fresh and final dry products of each sample were recorded and the percentage of moisture was calculated (Table 1).
Animals and diets: Twenty male Sprague-Dawley rats (weighing 180±9 g) were obtained from the animal house of the National Research Center, Giza, Egypt. The hypercholesterolemic diets were prepared from an AIN-93M purified rodent diet that conformed to the nutrient requirements for the maintenance of adult rats, set out by the National Research Council (Reeves et al., 1993) and supplemented with 0.25% of cholesterol. The current study protocol was approved by the Ethics Committee at National Research Center for animal study and the protocol conformed to the guidelines of the National Institutes of Health (National Research Council, 1996).
| Table 2: | Composition of diets fed to rat for 4 weeksa |
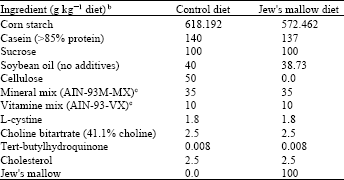 | |
aFormulated according to AIN-93 for rodent diet (Reeves et al., 1993). bThe total energy % as carbohydrates, protein and fat were about 74, 15, 10%, respectively versus 75, 15, 10%, respectively in the control group. cPrepared and mixed according to the method of the American Institute of Nutrition Rodent Diets (Reeves et al., 1993). Difference in the quantity added of starch, casein and oil in the different diets are adjusted, so that the final starch, casein and oil in all diets are the same | |
Experimental design: The rats were individually housed in metabolic cages kept in a room maintained at 22°C and subjected to a 12 h light-dark cycle with access to food from 4:00 pm to 8:00 am They were randomized to two groups and fed ad libitum commercial diet for 3 days, followed by the experimental diets for 4 weeks (Table 2). The diets were dispensed as a dry powder.
The two diets supplemented with 0.25% of cholesterol were isoenergetic and well equilibrated in terms of main nutrients. The supply of minerals and vitamins was reduced by 3.5-18.8% assuming that the Jew’s mallow dish already contained these elements. Cellulose (5%) was added to the control diet (hypercholesterolemic diet) as a DF source. Because Jew's mallow contained approximately 46% of total DF, 10% of the freeze-dried Jew's mallow powder was added to the experimental diet to provide a similar amount of DF. Samples of feces were collected during the last 3 days of the experimental period. Daily food consumption was noted and body weight was recorded twice a week.
Sampling procedures: The rats were anesthetized during the post-absorptive period (between 08:00 am and 11:00 am), when the cecal fermentation process is still active. Blood was drawn from the abdominal aorta into heparinzed tubes and centrifuged at 4000 rpm for 10 min. Plasma samples were stored at -40°C for lipid analysis. The cecum (wall with contents) was removed and weighed. The cecal contents were collected into microfuge tubes, immediately frozen (with liquid nitrogen) and stored at -40°C. For the SCFA analysis, supernatants were obtained by centrifuging the microtubes at 20,000 g, for 10 min at 4°C. The cecal wall was flushed clean with saline (0.9% NaCl), dried with filter paper and weighed (cecal wall weight). The liver was freeze clamped and stored at -40oC for the measurement of lipid contents.
Analytical procedures: The freeze-dried sample of Jew's mallow was ground in a Willy mill to 60 mesh size. Its moisture, crude protein (N X 6.25) and crude fat contents were determined by the standard methods of AOAC (1990). The carbohydrate content as nitrogen-free-extract was calculated by difference.
Determination of dietary fiber: Total, soluble and insoluble dietary fiber contents were analyzed using the enzymatic-gravimetric method (Lee et al., 1992). The method involves enzymatic hydrolysis with α-amylase, protease and amyloglucosidase, using the MES–TRIS buffer (all obtained from Sigma Chemical and included as part of the total dietary fiber kit, Cat. No. TDF-100A). The Jew's mallow sample was previously dried and rendered fat-free. The triplicate comprising approximately 0.5 g of a sample were suspended in 40 mL MES-TRIS buffer and subjected to an enzymatic hydrolysis sequence: 50 μL of thermoresistant α-amylase, in a water bath for 35 min and 100 μL of protease in a water bath at 60± 1°C for 30 min. After that, the pH was corrected to a range of 4.0-4.7 and 300 μL of amyloglucosidase, placed in a water bath at 60°C ± 1°C for 30 min, was added. After the precipitation of the soluble fiber with ethanol (95% v/v) at 60°C, the sample was filtered in fritted (sintered) glass crucibles (of the gooch type) using glass wool as filtering medium. The crucibles containing the residue were dried in an oven at 105°C and thereafter cooled in a desiccator before being weighed.
Determination of minerals: Sodium and potassium were determined by flame photometry while phosphorus was determined by the vanadomolybdate method (AOAC, 1990). The other minerals (Mg, Ca, Zn, Mn, Fe and Cu) were determined using an atomic absorption spectrophotometer. A 0.5 g portion of the Jew's mallow sample was digested on the Ethos Plus microwave workstation with computer-controlled easywave software (Milestone, CT, USA). The labstation provides 100% reliable quality assurance of the analytical sample process through its quality pressure chemical sensor, vent and reseal vessel technology. Digestion procedures were adapted from the Milestone Ethos-PLUS microwave labstation manual. A Perkin-Elmer (Analyst 100-Perkin-Elmer Inc., USA) atomic absorption spectrophotometer was used to analyze Mg, Ca, Zn, Mn, Fe and Cu in the digested samples. For confirming the precision and accuracy of the method, a standard reference material (SRM 1846) was analyzed (National Institute of Standards and Technology, Gaithersburg, MD, USA).
Determination of Short Chain Fatty Acids (SCFA): The SCFA were analyzed using a slight modification of the Gas-Liquid Chromatography (GLC) method of Remesy and Demigne (1974). SCFA were measured by GLC on aliquots of supernatants of cecal contents (20,000 g, 10 min at 4°C) after ethanolic extraction. To precipitate protein and extract SCFA, ~2-3 vols of ice-cold 100% ethanol were added to samples (450-700 μL) combined with 0.1 μmol of isobutyric acid internal standard. After centrifugation (10 min, 13,000 g, 4°C), the supernatant fractions were transferred to microcentrifuge tubes containing 6 μmol of NaOH, yielding a final pH of ~8. It was then evaporated in a very slight air current at 20°C. Just before GLC analysis, samples were redissolved in 80 μL of ice-cold 6.25% metaphosphoric acid and clarified by centrifugation (15 min, 4,000 g, 4°C) through 0.45 μm nylon microcentrifuge tube filters (Lida Manufacturing, Kenosha, WI). Samples (2 μL) were injected into the gas chromatograph, which employed the following conditions: a Supelco (Bellefonte, PA) GP 10% SP-100/1% H3PO4 on 80/100 Chromosorb WAW column (6 ftx4 mm ID), N2 carrier (73 mL min-1), injector temperature 175°C, oven temperature 125°C and a Flame-Ionization Detector (FID) set at 180°C. Conversion factors (μM/FID response peak area) and assay linearity were determined daily from authentic SCFA mixed standards (fatty acid chain lengths C2-C5, including isobutyric acid) covering the range of sample concentrations. Water blanks were chromatographed between each sample. The coefficient of variation of duplicate sample analyses averaged 6.1±0.5%.
Determination of Bile acids and neutral steroids: Bile acids and neutral steroids were extracted twice from feces at 70°C for 2 h by 2x10 volumes of alkaline ethanol (KOH 0.5 mol L-1). Bile acids were quantified using a reaction catalyzed by 3α-hydroxysteroid dehydrogenase (EC 1.1.1.50; Cat. No. H1506 Sigma-Aldrich Chemical Co.) (Turley and Dietschy, 1978). Neutral steroids in alkaline ethanol solution (100 μL) were extracted three times with hexane (500 μL) after addition of 5 α-cholestane (internal standard, Cat. No. R205370 Sigma-Aldrich Chemical Co.). The hexane extract was concentrated to 200 and 2 μL of it were injected into the gas chromatograph (Perkin Elmer auto system XL) fitted with a 12 mx0.25 mm fused silica capillary column (BP 10) and a flame-ionization detector. Helium was used as carrier gas and an isocratic temperature (260°C) was used for the steroid separation. Sterol concentrations were calculated from the peak area relative to the area of the internal standard.
Determination of plasma and liver lipids: Plasma total cholesterol concentrations (Cat. No. CH 200), HDL (Cat. No. CH 203) and triglyceride (Cat. No. TR 1575) concentrations were enzymatically determined using a kit purchased from Randox Laboratories, Antrin, UK. Plasma lipoproteins (LDL+VLDL) were calculated as follows:
LDL+VLDL cholesterol = Total cholesterol–HDL cholesterol |
The atherogenic index was calculated as:
Atherogenic index = (Total cholesterol–HDL–Cholesterol)x HDL–cholesterol-1 (Muramatsu et al., 1986) |
Liver lipids were extracted with chloroform/methanol (2:1, v/v) according to the method previously described (Mazur et al., 1990). Triglycerides in lipid residue were saponified by 0.5 mol L-1 KOH-ethanol at 70°C for 30 min, 0.15 mol L-1 MgSO4 was then added to neutralize the mixture. Cholesterol in the lipid residue was measured with the enzymatic procedure as described earlier. A polyvalent control serum (Biotrol-33 plus, lot No. 577; Merck-Chemical Co.) was treated in parallel with the samples and served as a control of the accuracy of results in triglyceride and cholesterol analysis.
Statistical analysis: Values were noted as the Means±SEM and the differences between the two groups were determined by the Student’s t-test for the significance of the difference between the means. Differences of p<0.05 were considered significant (Rao and Chakraborty, 1991).
Food intake, body and organ weight and digestive fermentation: There was no difference detected between the two groups either in the daily food intake (18.5±1.0 g day-1 in control vs. 20.2±1.2 g day-1 in rats fed Jew's mallow diet) or in the final body weight (281.8±7.9 g in control vs. 289.8±8.3 g) (Table 3).
The incorporation of 10% of freeze-dried Jew's mallow significantly affected the relative liver weight (4.6±0.1 in control vs. 4.1±0.1 g) of rats fed Jew's mallow diet, they were significantly smaller than those of control (p<0.05).
The cecal wall weight was found to have significantly increased from 0.80±0.09 g in control rats to 1.32±0.08 g (+65%, p<0.05) in rats fed Jew's mallow diet. A three-fold increase of SCFA pool in the caecum (Fig. 1) was observed resulting in a significant rise (p<0.05) of all SCFAs (+294% for acetate, +205% for propionate and +144% for butyrate).
Plasma and tissue lipids: The supplementation with 0.25% of cholesterol in the diet induced a substantial accumulation of plasma cholesterol (2.8 mol L-1) and triglycerides (1.6 mol L-1) in control rats but not in rats fed Jew's mallow diet.
| Table 3: | Body weight, food intake and feed efficiency in rats control and fed Jew's mallow diets for 4 weeksa |
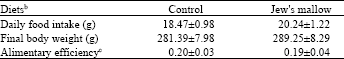 | |
| aValues are Mean±SEM, n = 10. bDiets were supplemented with 0.25% cholesterol. cAlimentary efficiency = Daily weight gain/Daily food intake. The other explanations as in Table 4 | |
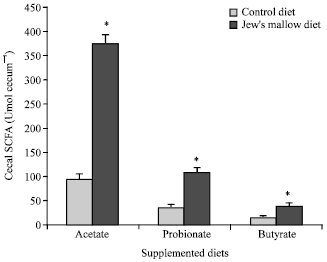 | |
| Fig. 1: | Cecal short-chain fatty acids in rats fed a control or a 10% Jew's mallow supplemented diets for 4 week. *Differences were considered statistically significant for p<0.05. The other explanations as in Table 4 |
The Jew's mallow diet led to a slight but significant lowering (p<0.05) of plasma cholesterol and reduced significantly (p<0.05) the atherogenic index for 49% with respect to the control group. Liver cholesterol was significantly lowered in rats fed Jew's mallow diet (-38%) vs. control rats (Table 4).
Cholesterol intake and digestive balance of bile acids: As shown in Table 4, the daily cholesterol intake was similar in both groups. However, the daily consumption of Jew's mallow induced a greater significant fecal excretion of neutral sterols (+72%) and especially coprostanol (+79%) compared to a 67% increase of cholesterol excretion. Digestive neutral sterols balance was thus significantly altered by Jew's mallow diet and resulted in a decrease of apparent cholesterol absorption.
Table 4 shows a significant increase in the percentage of apparently absorbed cholesterol excreted in feces as bile acids, leading to a significant decrease of total steroid balance in rats fed Jew's mallow diet.
| Table 4: | Effect of ingestion of Jew's mallow diet for 4 weeks on cholesterol metabolism in rats |
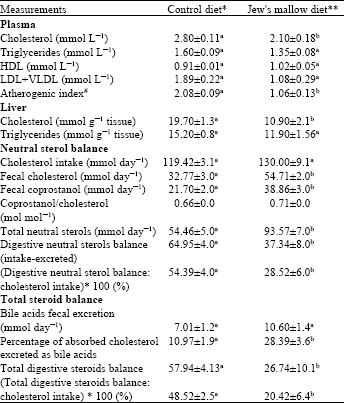 | |
Values are Means±SEM, n = 10. Values in the same raw that do not share the same superscript are significantly different. Differences were considered statistically significant for p<0.05. Comparisons between the two groups were analyzed using the Student t-test. #Atherogenic Index = (Total cholesterol–HDL–Cholesterol)xHDL-Cholesterol-1. *The control diet contained 5% cellulose as DF. **The test diet was supplemented with 100 g of freeze-dried Jew's mallow kg-1 (providing approximately 5% DF) | |
The ratio of total digestive steroid balance to cholesterol intake, which represents the apparent absorption of dietary cholesterol, was significantly depressed by the Jew's mallow diet, representing only 20% of the cholesterol intake vs. 48% in the control diet (p<0.05).
Experimental model description: The non-energetic moiety of vegetables (fibers, minerals, micronutrients) is complex and plays an important role in mediating various health effects such as cardiovascular diseases (Ness and Powles, 1997). Numerous studies have investigated separately the effects of fibers on cholesterol metabolism and prevention of cardiovascular diseases; however, little is known about the mechanism of action of complex plant foods such as Jew's mallow.
After documenting the micronutrient contents in several cultivars. Jew's mallow differs from other vegetables in that it contains several types of micronutrients and fibers (Guptaa et al., 2005; Hadjichambis et al., 2008; Innami et al., 2005; Oboh et al., 2009; Oboh and Akindahunsi, 2004; Shrif et al., 1980; Thrichopoulou et al., 2000; Zeghichi et al., 2003). Jew's mallow provide the best opportunity for the study of complex plant foods, rich in dietary fiber content, on cardiovascular diseases.
In this study, normal rats were used to investigate the potential hypocholesterolemic effect of a supplemented cholesterol diet rich in Jew's mallow. Rat strains provide the best opportunity for the study of both abnormal metabolism and pathophysiology leading to cardiovascular diseases (Russell and Proctor, 2006). Despite the limitations of the rat model, rats can synthesize vitamin C, their carotenoid bioavailability is lower than in humans, they can absorb dietary cholesterol better than humans and they are often used to evaluate the effects of dietary products on serum cholesterol concentrations (Muramatsu et al., 1986; Levrat et al., 1993; Tebib et al., 1994). The rats accumulated cholesterol, at any level of cholesterol intake, in the liver and plasma. Conversion of cholesterol to bile acids, catalyzed by hepatic 7α-hydroxylase, is the major regulated pathway by which cholesterol is removed from the body. Dietary cholesterol completely blocked the suppressive effects of bile acids on 7α-hydroxylase expression in the rat. Indeed the highest levels of expression were observed in animals fed cholesterol plus cholic acid. Since, cholic acid promotes cholesterol absorption, it greatly increased the cholesterol content of the liver (Horton et al., 1995).
The present results showed that diet containing leaf vegetable (Jew's mallow) produces some interesting digestive- and metabolic responses in these animals. Since the rat is a low-response animal, we chose to investigate with a high level of Jew's mallow in the diet to obtain a significant response. Moreover, the supplementation of the diet with 0.25% of cholesterol for four weeks allowed the development of a significant hypercholesterolemia without inducing fatty liver (Lecumberri et al., 2007; Nicolle et al., 2004). In these experimental conditions, the Jew's mallow diet elicited a slight but significant cholesterol-lowering effect in plasma and tended to a significant atherogenic index-decreasing effect. Therefore, present data support a clear cholesterol-lowering action of Jew's mallow. However, results relating to cholesterol-lowering effects of Jew's mallow in humans are lacking. Such an effect can be considered as beneficial for cardiovascular disease prevention and treatment. A similar effect was observed in earlier study with high-fiber diets or dried lettuce. Fibers are known to recover the lipoprotein profile in cholesterol-fed rats (Nicolle et al., 2004). This effect is also in keeping with earlier investigations on hamsters and rats, showing that apples, being rich in soluble fibers, were able to lower cholesterol in the most atherogenic lipoproteins (VLDL/LDL). However, the quantity of fiber provided by added vegetables is unlikely to explain the totality of their effects (Chau et al., 2004; Thrichopoulou et al., 2000). The exact mechanism is not yet clearly understood and this point needs further investigation.
Dietary fibers, cholesterol absorption and metabolism: The cholesterol-lowering effects of Jew's mallow may be ascribed to the fiber fraction. A diet containing 10% Jew's mallow (with 45% of total dietary fiber content) corresponds to a 4.6% dietary fiber, which is relatively low for the rats when compared to control rats (fed 5% dietary fiber as cellulose). Jew's mallow fibers (comprising only 38% of soluble fibers) are mainly represented by poorly lignified primary cell walls, rich in soluble hemicellulose and pectin and highly fermentable fibers (Bourquin et al., 1993; Guptaa et al., 2005). The impact of pectin, or various soluble fibers, on lipid metabolism has been well established both in humans and animal models (Aller et al., 2004; Levrat et al., 1993).
Dietary fibers can also exert hypocholesterolemic effects by increasing fecal excretion of total steroids (neutral sterols and bile acids) (Sembriesa et al., 2004). Although, the hypocholesterolemic action of Jew's mallow has not been established in human subjects, present study indicated that the Jew's mallow diet lowered the digestive absorption of cholesterol in the rat. The mechanisms of inhibition of cholesterol absorption have been well documented; they include mainly disturbance of micelle formation and slowing of cholesterol transfer to the brush border across the unstirred layer (Stedronsky, 1994 ). Jew's mallow ingestion also increased the amount of dietary cholesterol excreted in feces as bile acids. This could reflect an entrapment of bile acids within the viscous medium, as well as an accelerated billary influx (Moundas et al., 1997). Such effects on steroids balance are consistent with a lower accumulation of dietary cholesterol in blood and tissues. It is noteworthy that the Jew's mallow diet exhibited a greater effect on hepatic cholesterol than on plasma cholesterol. It is unlikely that the small decrease in the energetic density of the Jew's mallow diet (-5.4%) could exert such a magnified effect. This may rather be the result of both, a decrease of digestive cholesterol absorption and a higher synthesis of bile acids. In humans, both effects may contribute to individual differences in responsiveness to a high cholesterol diet (Horton et al., 1995).
On the other hand, a cholesterol-enriched diet is known to inhibit the HMG-CoA reductase activity (Levrat-Verny et al., 2000). It is likely this activity could be maintained at a low level with the Jew's mallow diet. Fibers could also exert indirect effects on cholesterol metabolism. Their fermentation in the large intestine leads to a production of SCFA such as propionate, the most effective cholesterol-lowering agent (Jenkins et al., 2000).
In the present study, we observed a 205% rise of propionic acid in the cecal pool. In numerous studies on fiber fermentation and SCFA metabolism, a direct relation between SCFA cecal pool and SCFA absorption was observed (Nicolle et al., 2004). From early studies of isolated hepatocytes, it was postulated that propionate has an inhibitory effect on cholesterol metabolism (Demigne et al., 1995). This hypothesis received some support from observations that propionate can significantly inhibit cholesterol biosynthesis from acetate in hepatocytes (Wright et al., 1990). Nevertheless, the effectiveness of propionate on cholesterol metabolism in the liver is likely to be less than the direct effect of fiber on digestive cholesterol absorption or its indirect effect on the cholesterol conversion into bile acids. Altogether, such mechanisms are able to reduce plasma cholesterol concentration.
The most important finding of this study is that the Jew's mallow showed an important hypocholesterolemic action, in vivo, by its fiber content and possibly by the action various other micronutrients such as phytosterols. Although, we are still on the path to isolate and identify the active compound(s), flavonoids is one of the possible candidates of the active compound(s) in Jew's mallow. Indeed, Jew's mallow abundantly contains 5-caffeoylquinic acid, 3, 5-dicaffeoylquinic acid, quercetin 3-galactoside, quercetin 3-glucoside, quercetin 3- (6-malonylglucoside), quercetin 3-(malonylgalactoside), ascorbic acid, α-tocopherol, chlorophyll, etc. and the content of quercetin glycosides is remarkable (Azuma et al., 1999). In earlier reports, it has been demonstrated that several phytosterols, such as flavonoids improve the lipidemic profile (Levrat et al., 1993; Muramatsu et al., 1986; Nicolle et al., 2004; Tebib et al., 1994). These reports indicate that an intake of vegetables, fruits and tea have the potential to reduce cholesterol, since these foodstuffs abundantly contain dietary fiber and/or various natural phytosterols. Therefore, Jew’s mallow is the attractive foodstuff for isolation and identification of a phytosterols that are able to act in vivo. Further research is needed to investigate the effect of diet containing Jew's mallow on antioxidant status including studies on humans to elucidate the mechanisms of action.
The consumption of leaf vegetable (Jew's mallow) with a hypercholesterolemic diet improved the lipidemic profile and increased the total cholesterol end-products excretion, suggesting that Jew's mallow might contribute to a reduction of cardiovascular risk.
This study was supported by National Research Center, Giza, Egypt (Grant No. 8040440). I thank M. Farag for his technical assistance and M. Ali and O. Watkins for their editorial assistance.