Research Article
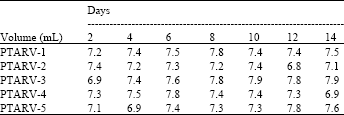
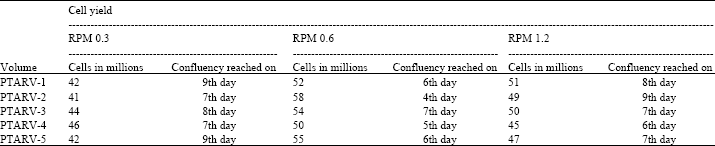
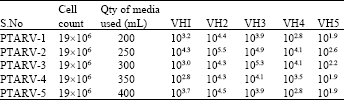
Standardization and Assessment of Cell Culture Media Quantities in Roller Poly Ethylene Terephthalate Bottles Employed in the Industrial Rabies Viral Vaccine Production
Pasteur Institute of India, Coonoor, The Nilgiris, India
S. Chaansha
Kongunadu college of Arts and Science, Coimbatore, India
K. Rajesh
Muthayammal College of Arts and Science, Rasipuram, India
T. Santhiya
Kongunadu college of Arts and Science, Coimbatore, India
C. Charles
Pasteur Institute of India, Coonoor, The Nilgiris, India
K.N. Venkataramana
Pasteur Institute of India, Coonoor, The Nilgiris, India