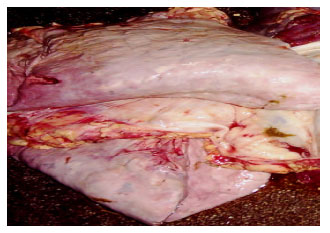
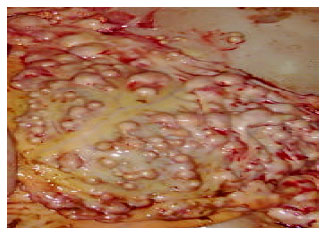
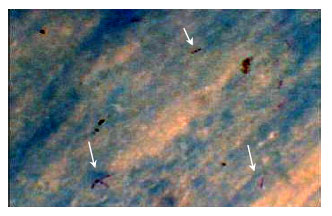
Research Article
Preliminary Investigation of Bovine Tuberculosis in Suspected Beef from a Metropolitan Abattoir in Ghana with Ziehl-Neelsen Microscopy
Applied Radiation Biology Centre, Radiological and Medical Sciences Research Institute, Ghana Atomic Energy Commission, P.O. Box LG 80, Legon, Accra, Ghana
E.E. Mak-Mensah
Department of Biochemistry and Biotechnology, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
D.G. Achel
Applied Radiation Biology Centre, Radiological and Medical Sciences Research Institute, Ghana Atomic Energy Commission, P.O. Box LG 80, Legon, Accra, Ghana
O.K. Gyamfi
Cellular and Clinical Research Centre, Radiological and Medical Sciences Research Institute, Ghana Atomic Energy Commission, Accra, Ghana
K.D. Bedzra
Cellular and Clinical Research Centre, Radiological and Medical Sciences Research Institute, Ghana Atomic Energy Commission, Accra, Ghana