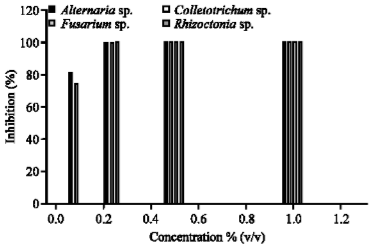
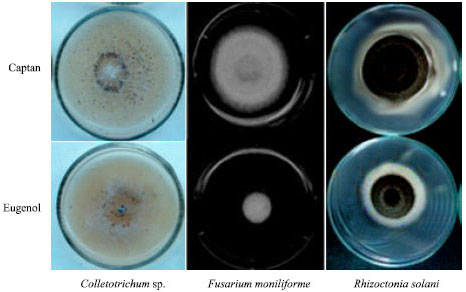
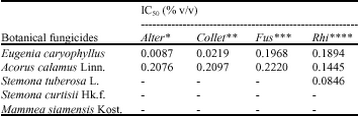
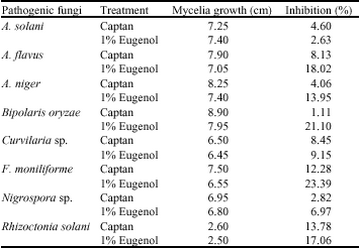
Research Article
Implementation of Bio-Fungicides and Seed Treatment in Organic Rice cv. KDML 105 Farming
Department of Agricultural Technology, Faculty of Technology, Mahasarakham University, Talard Sub-District, Muang District, Maha Sarakham 44000, Thailand