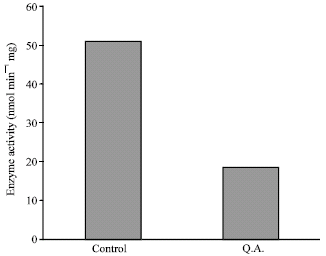
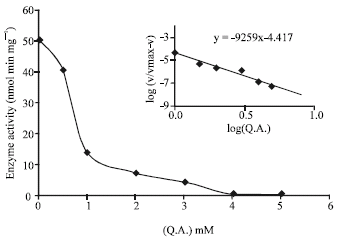
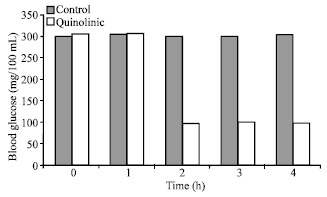
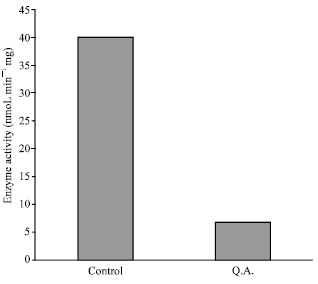
Research Article
New Evidence on Hypoglycemic Effect of Quinolinic Acid in Diabetic Rats
Department of Biology, Faculty of Sciences, Shahid Chamran University of Ahvaz, Ahwaz, Iran
I. Safari
Department of Biology, Faculty of Sciences, Shahid Chamran University of Ahvaz, Ahwaz, Iran
M.S. Dayer
Department of Parasitology and Medical Entomology,Tarbiat Modares University, Tehran, Iran