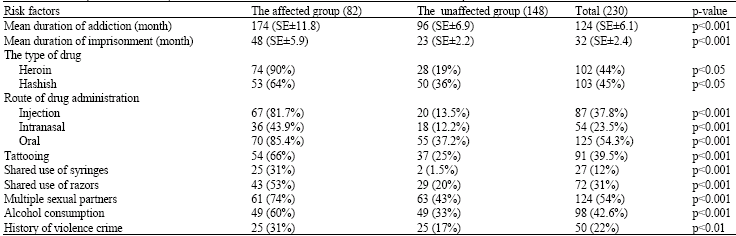
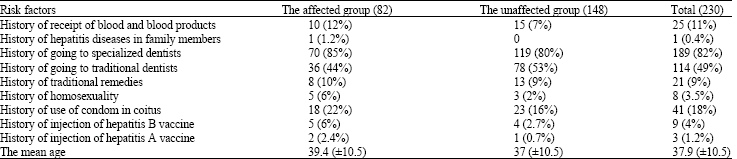
Research Article
Seroprevalence of Hepatitis C Infection and Associated Risk Factors among Addicted Prisoners in Sari-Iran
Department of Community Health Nursing,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
F. Salmeh
Department of Community Health Nursing,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
T. Yaghoobi
Department of Nursing Management,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
M. Yaghoubian
Department of Nursing Management,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
M.B. Nesami
Department of Medical Surgical Nursing,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
Z. Esmaeeli
Department of Pediatric Nursing,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
N. Vaezzadeh
Department of Pediatric Nursing,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
S. Shahmohammadi
Department of Nursing and Midwifery Research,Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
S. Modanloo
Department of Psychiatric Nursing, Nasibeh Nursing and Midwifery College, Mazandaran University of Medical Sciences, Sari, Iran
A.A. Sadeghian
Department of CDC, Mazandaran University of Medical Sciences, Sari, Iran
S.J. Abdolmanafi
Sari, Mazandaran Province, Iran
R.A. Mohammadpour
Department of Biostatistics, Faculty of Health, Mazandaran University of Medical Sciences, Sari, Iran
H. Siamian
Department of Medical Records, Faculty of Paramedical Sciences, Mazandaran University of Medical Sciences, Sari, Iran
A. Khosravi
Department of Anesthesiology, Faculty of Medicine, Yasouj University of Medical Sciences, Yasouj, Iran