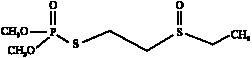Research Article
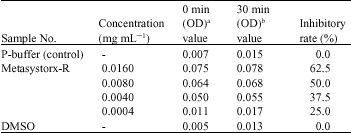
Effect of Metasystox-R on Marine Nitrosomonas sp. as a Nitrification Inhibitor
Department of Marine Biology, Islamic Azad University, North Tehran Branch, P.O. Box 19375-166, Tehran, Iran
Maryam Amirsharifi
Department of Marine Biology, Islamic Azad University, North Tehran Branch, P.O. Box 19375-166, Tehran, Iran
Akbar Esmaeili
Department of Marine Biology, Islamic Azad University, North Tehran Branch, P.O. Box 19375-166, Tehran, Iran
Lida Salimi
Department of Marine Biology, Islamic Azad University, North Tehran Branch, P.O. Box 19375-166, Tehran, Iran